Home
/
PHYSICS
/
FORM FOUR PHYSICS STUDY NOTES TOPIC 3: RADIOACTIVITY & TOPIC 4: THERMIONIC EMISSION
FORM FOUR PHYSICS STUDY NOTES TOPIC 3: RADIOACTIVITY & TOPIC 4: THERMIONIC EMISSION
LINK OF OTHER SUBJECTS >>>>>
TOPIC 3: RADIOACTIVITY
The Nucleus of an Atom
The Structure of the Nucleus of an Atom
Describe the structure of the nucleus of an atom
The
word atom is derived from the Greek word atom which means indivisible.
The Greeks concluded that matter could be broken down into particles to
small to be seen. These particles were called atoms.
Atoms
are composed of three type of particles: protons, neutrons, and
electron.Protons and neutrons are responsible for most of the atomic
mass e.g in a 150 person 149 lbs, 15 oz are protons and neutrons while
only 1 oz. is electrons.The mass of an electron is very small (9.108 X
10-28 grams).
Both
the protons and neutrons reside in the nucleus. Protons have a positive
(+) charge, neutrons have no charge i.e they are neutral. Electrons
reside in orbitals around the nucleus. They have a negative charge (-).
It
is the number of protons that determines the atomic number, e.g., H =
1. The number of protons in an element is constant (e.g., H=1, Ur=92)
but neutron number may vary, so mass number (protons + neutrons) may
vary.
The same element may contain varying numbers of neutrons; these forms of an element are called isotopes. The chemical properties of isotopes are the same, although the physical properties of some isotopes may be different.
Some
isotopes are radioactive-meaning they "radiate" energy as they decay to
a more stable form, perhaps another element half-life: time required
for half of the atoms of an element to decay into stable form. Another
example is oxygen, with atomic number of 8 can have 8, 9, or 10
neutrons.
The Atomic Number, Mass Number and Isotopes of an Element and their Symbols
Explain the atomic number, mass number and isotopes of an element and their symbols
The
atomic number of a chemical element (also known as its proton number)
is the number of protons found in the nucleus of an atom of that
element.Therefore it is identical to the charge number of the nucleus.
It is conventionally represented by the symbol Z.
The
atomic number uniquely identifies a chemical element. In an uncharged
atom, the atomic number is also equal to the number of electrons.
The atomic number, Z, should not be confused with the mass number, A.
Mass
number is the number of nucleons, i. e the total number of protons and
neutrons in the nucleus of an atom. ---The number of neutrons, N, is
known as the neutron number of the atom; thus, A = Z + N (these
quantities are always whole numbers).
Since
protons and neutrons have approximately the same mass (and the mass of
the electrons is negligible for many purposes) and the mass defect of
nucleon binding is always small compared to the nucleon mass, the atomic
mass of any atom, when expressed in unified atomic mass units (making a
quantity called the "relative isotopic mass"), is roughly (to within
1%) equal to the whole number A.
Isotopes
Isotopes are atoms with the same atomic number Z but different neutron numbers N, and hence different atomic masses.
A
little more than three-quarters of naturally occurring elements exist
as a mixture of isotopes (see monoisotopic elements), and the average
isotopic mass of an isotopic mixture for an element (called the relative
atomic mass) in a defined environment on Earth, determines the
element's standard atomic weight.
Historically,
it was these atomic weights of elements (in comparison to hydrogen)
that were the quantities measurable by chemists in the 19th century.The
chemical properties of isotopes are the same, although the physical
properties of some isotopes may be different.
Some
isotopes are radioactive-meaning they "radiate" energy as they decay to
a more stable form, perhaps another element half-life: time required
for half of the atoms of an element to decay into stable form. Another
example is oxygen, with atomic number of 8 can have 8, 9, or 10
neutrons.
Forces Holding the Nucleus
Mention forces holding the nucleus
Stable and unstable atoms
There
are forces within the atom that account for the behavior of the
protons, neutrons, and electrons. Without these forces, an atom could
not stay together.
Recall
that protons have a positive charge, electrons a negative charge, and
neutrons are neutral. According to the laws of physics, like charges
repel each other and unlike charges attract each other. A force called
the strong force opposes and overcomes the force of repulsion between
the protons and holds the nucleus together.
The
net energy associated with the balance of the strong force and the
force of repulsion is called the binding energy. The electrons are kept
in orbit around the nucleus because there is an electromagnetic field of
attraction between the positive charge of the protons and the negative
charge of the electrons.
In
some atoms, the binding energy is great enough to hold the nucleus
together. The nucleus of this kind of atom is said to be stable. In some
atoms the binding energy is not strong enough to hold the nucleus
together, and the nuclei of these atoms are said to be unstable.
Unstable atoms will lose neutrons and protons as they attempt to become
stable.
- Binding energy is the net energy that is the result of the balance with the strong force and the repulsive force, and this is the amount of energy that holds the nucleus together.
- A stable atom is an atom that has enough binding energy to hold the nucleus together permanently.
- An unstable atom does not have enough binding energy to hold the nucleus together permanently and is called a radioactive atom.
Natural Radioactivity
The Concept of Radioactivity
Explain the concept of radioactivity
Radioactive
decay, also known as nuclear decay or radioactivity, is the process by
which a nucleus of an unstable atom loses energy by emitting ionising
radiation.
A
material that spontaneously emits such radiation — which includes alpha
particles, beta particles, gamma rays and conversion electrons — is
considered radioactive.
Radioactive
decay is a stochastic (i.e. random) process at the level of single
atoms, in that, according to quantum theory, it is impossible to predict
when a particular atom will decay.
The
chance that a given atom will decay never changes, that is, it does not
matter how long the atom has existed. For a large collection of atoms
however, the decay rate for that collection can be calculated from their
measured decay constants or half-lives. The half-lives of radioactive
atoms have no known limits for shortness or length of duration, and
range over 55 orders of magnitude in time.
Properties of the Radiations Emitted by Radio-active Substances
Describe properties of the radiations emitted by radio-active substances
There
are many types of radioactive decay . A decay, or loss of energy,
results when an atom with one type of nucleus, called the parent
radionuclide (or parent radioisotope), transforms into an atom with
anucleus in a different state, or with a nucleus containing a different
number of protons and neutrons. The product is called the daughter
nuclide. In some decays, the parent and the daughter nuclides are
different chemical elements, and thus the decay process results in the
creation of an atom of a different element. This is known as a nuclear
transmutation.
The Nuclear Changes due to the Emission of Alpha ('8c'b1), Beta ('8cuc0u8804 ) and Gamma ('8cu8805 ) Radiations
Explain the nuclear changes due to the emission of Alpha ('8c'b1), Beta ('8cuc0u8804 ) and Gamma ('8cu8805 ) radiations
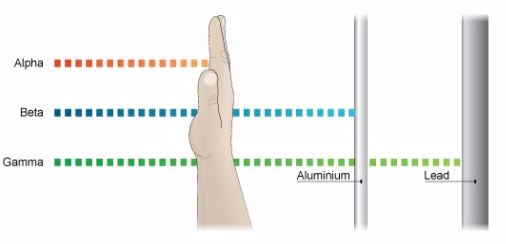
Properties of Alpha Rays
- Alpha rays or alpha particles are the positively charged particles.
- Alpha particles have the least penetration power. They cannot penetrate the skin but this does not mean that they are not dangerous.
- Since they have a great ionisation power, so if they get into the body they can cause serious damage. They have the ability of ionising numerous atoms a short distance. It is due to this reason that the radioactive substance that releases alpha particles needs to be handled with rubber gloves. It should not be inhaled, eaten or allowed near open cuts.
Properties of Beta Rays.
- Beta particles are highly energetic electrons which are released from inside of a nucleus.
- They are negatively charged and have a negligible mass.
- Beta particles have a greater penetration power than the alpha particles and can easily travel through the skin.
- Though beta particles have less ionisation power than the alpha particles but still they are dangerous and so their contact with the body must be avoided.
Properties of Gamma Rays
- They have greatest power of penetration.
- They are the least ionizing but most penetrating and it is extremely difficult to stop them from entering the body.
- These rays carry huge amount of energy and can even travel through thin lead and thick concrete.
The Detection of '8c'b1, '8cuc0u8804 and '8cu8805 Radiations
Explain the detection of '8c'b1, '8cuc0u8804 and '8cu8805 radiations
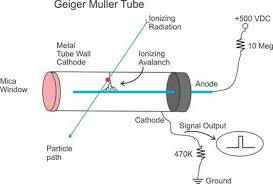
Geiger Counter, with Geiger-Mueller (GM) Tube or Probe
A
GM tube is a gas-filled device that, when a high voltage is applied,
creates an electrical pulse when radiation interacts with the wall or
gas in the tube. These pulses are converted to a reading on the
instrument meter.
If
the instrument has a speaker, the pulses also give an audible click.
Common readout units are roentgens per hour (R/ hr), milliroentgens per
hour (mR/hr), rem per hour (rem/hr), millirem per hour (mrem/hr), and
counts per minute (cpm).
GM
probes (e.g., "pancake" type) are most often used with handheld
radiation survey instruments for contamination measurements. However,
energy-compensated GM tubes may be employed for exposure measurements.
Further,
often the meters used with a GM probe will also accommodate other
radiation-detection probes. For example, a zinc sulfide (ZnS)
scintillator probe, which is sensitive to just alpha radiation, is often
used for field measurements where alpha-emitting radioactive materials
need to be measured.
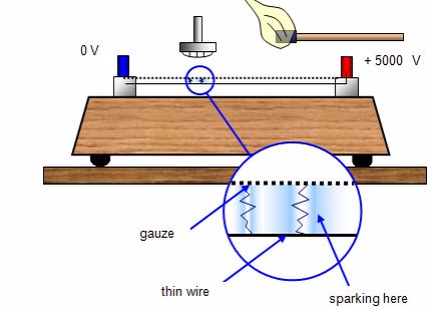
Spark counter
This
consists of a fine metal gauze mounted about a millimetre away from a
thin wire.A voltage is applied between the two so that sparking takes
place between them - this usually requires some 4000 - 5000 V. The
voltage is then reduced until sparking just stops.
If
an alpha-source is brought up close to the gauze it will ionise the
air, and sparks will occur between the gauze and wire. With beta and
gamma sources insufficient ions are usually produced for sparking to
take place.The spark counter can be used to measure the range of
alpha-particles.
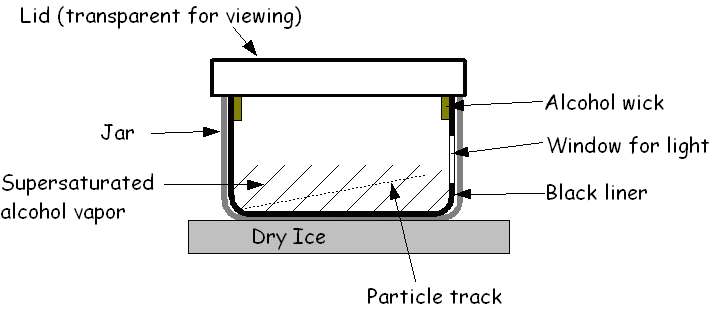
Cloud chamber
The cloud chamber, also known as the Wilson chamber, is a particle detector used for detecting ionising radiation.
Rare
picture shows in a single shot the 4 particles that we can detect in a
cloud chamber: proton, electron, muon (probably) and alpha. In its most
basic form, a cloud chamber is a sealed environment containing a
supersaturated vapor of water or alcohol.
When
a charged particle (for example, an alpha or beta particle) interacts
with the mixture, the fluid is ionized. The resulting ions act as
condensation nuclei, around which a mist will form (because the mixture
is on the point of condensation).
The
high energies of alpha and beta particles mean that a trail is left,
due to many ions being produced along the path of the charged particle.
These tracks have distinctive shapes (for example, an alpha particle's
track is broad and shows more evidence of deflection by collisions,
while an electron's is thinner and straight).
When
any uniform magnetic field is applied across the cloud chamber,
positively and negatively charged particles will curve in opposite
directions, according to the Lorentz force law with two particles of
opposite charge.
Other devices used to detect radiation include:
- Photographic film
- Bubble chamber
- Gold-leaf electroscope
Half-Life as Applied to a Radioactive Substance
Describe half-life as applied to a radioactive substance
Half
life can be defined as the time taken for the number of nuclei in a
radioactive material to halve. It can also be defined as the time taken
for the count rate of a sample of radioactive material to fall to half
of its starting level.
The
count rate is measured by using an instrument called a Geiger-Muller
tube over a period of time. A Geiger-Muller tube detects radiations by
absorbing the radiation and converting it into an electrical pulse which
triggers a counter and is displayed as a count rate.
The
release of radiation by unstable nuclei is called radioactive decay.
This process occurs naturally and cannot be influenced by chemical or
physical processes.
The
release of radiation is also a random event and overtime the activity
of the radioactive material decreases. It is not possible to predict
when an individual nucleus in a radioactive material will decay.
But
it is possible to measure the time taken for half of the nuclei in a
radioactive material to decay. This is called the half life of
radioactive material or radioisotope.
The Half-Life of a Radioactive Element
Determine the half-life of a radioactive element
An exponential decay process can be described by any of the following three equivalent formulas:

where
- N0 is the initial quantity of the substance that will decay (this quantity may be measured in grams, moles, number of atoms, etc).
- N(t) is the quantity that still remains and has not yet decayed after a time t.
- t1⁄2 is the half-life of the decaying quantity.
- τis a positive number called the mean lifetime of the decaying quantity.
- λis a positive number called the decay constant of the decaying quantity.
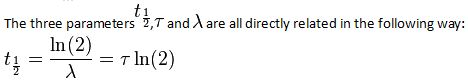
Where ln (2) is the natural logarithm of 2 (approximately 0.693).
By
plugging in and manipulating these relationships, we get all of the
following equivalent descriptions of exponential decay, in terms of the
half-life:

The Application of a Natural Radioactive Substances
Identify the applications of a natural radioactive Substances
Medical Uses
Hospitals,
doctors, and dentists use a variety of nuclear materials and procedures
to diagnose, monitor, and treat a wide assortment of metabolic
processes and medical conditions in humans. In fact, diagnostic x-rays
or radiation therapy have been administered to about 7 out of every 10
Americans. As a result, medical procedures using radiation have saved
thousands of lives through the detection and treatment of conditions
ranging from hyperthyroidism to bone cancer.
The
most common of these medical procedures involves the use of x-rays — a
type of radiation that can pass through our skin. When x-rayed, our
bones and other structures cast shadows because they are denser than our
skin, and those shadows can be detected on photographic film. The
effect is similar to placing a pencil behind a piece of paper and
holding the pencil and paper in front of a light. The shadow of the
pencil is revealed because most light has enough energy to pass through
the paper, but the denser pencil stops all the light. The difference is
that x-rays are invisible, so we need photographic film to "see" them
for us. This allows doctors and dentists to spot broken bones and dental
problems.
X-rays
and other forms of radiation also have a variety of therapeutic uses.
When used in this way, they are most often intended to kill cancerous
tissue, reduce the size of a tumor, or reduce pain. For example,
radioactive iodine (specifically iodine-131) is frequently used to treat
thyroid cancer, a disease that strikes about 11,000 Americans every
year.
X-ray
machines have also been connected to computers in machines called
computerized axial tomography (CAT) or computed tomography (CT)
scanners. These instruments provide doctors with color images that show
the shapes and details of internal organs. This helps physicians locate
and identify tumors, size anomalies, or other physiological or
functional organ problems.
In
addition, hospitals and radiology centers perform approximately 10
million nuclear medicine procedures in the United States each year. In
such procedures, doctors administer slightly radioactive substances to
patients, which are attracted to certain internal organs such as the
pancreas, kidney, thyroid, liver, or brain, to diagnose clinical
conditions.
Academic and Scientific Applications
Universities,
colleges, high schools, and other academic and scientific institutions
use nuclear materials in course work, laboratory demonstrations,
experimental research, and a variety of health physics applications. For
example, just as doctors can label substances inside people's bodies,
scientists can label substances that pass through plants, animals, or
our world. This allows researchers to study such things as the paths
that different types of air and water pollution take through the
environment. Similarly, radiation has helped us learn more about the
types of soil that different plants need to grow, the sizes of newly
discovered oil fields, and the tracks of ocean currents.
In
addition, researchers use low-energy radioactive sources in gas
chromatography to identify the components of petroleum products, smog
and cigarette smoke, and even complex proteins and enzymes used in
medical research.
Archaeologists
also use radioactive substances to determine the ages of fossils and
other objects through a process called carbon dating. For example, in
the upper levels of our atmosphere, cosmic rays strike nitrogen atoms
and form a naturally radioactive isotope called carbon-14. Carbon is
found in all living things, and a small percentage of this is carbon-14.
When a plant or animal dies, it no longer takes in new carbon and the
carbon-14 that it accumulated throughout its life begins the process of
radioactive decay. As a result, after a few years, an old object has a
lower percent of radioactivity than a newer object. By measuring this
difference, archaeologists are able to determine the object's
approximate age.
Industrial Uses
We
could talk all day about the many and varied uses of radiation in
industry and not complete the list, but a few examples illustrate the
point. In irradiation, for instance, foods, medical equipment, and other
substances are exposed to certain types of radiation (such as x-rays)
to kill germs without harming the substance that is being disinfected —
and without making it radioactive. When treated in this manner, foods
take much longer to spoil, and medical equipment (such as bandages,
hypodermic syringes, and surgical instruments) are sterilized without
being exposed to toxic chemicals or extreme heat. As a result, where we
now use chlorine — a chemical that is toxic and difficult-to-handle — we
may someday use radiation to disinfect our drinking water and kill the
germs in our sewage. In fact, ultraviolet light (a form of radiation) is
already used to disinfect drinking water in some homes.
Similarly,
radiation is used to help remove toxic pollutants, such as exhaust
gases from coal-fired power stations and industry. For example, electron
beam radiation can remove dangerous sulphur dioxides and nitrogen
oxides from our environment. Closer to home, many of the fabrics used to
make our clothing have been irradiated (treated with radiation) before
being exposed to a soil-releasing or wrinkle-resistant chemical. This
treatment makes the chemicals bind to the fabric, to keep our clothing
fresh and wrinkle-free all day, yet our clothing does not become
radioactive. Similarly, nonstick cookware is treated with gamma rays to
keep food from sticking to the metal surface.
The
agricultural industry makes use of radiation to improve food production
and packaging. Plant seeds, for example, have been exposed to radiation
to bring about new and better types of plants. Besides making plants
stronger, radiation can be used to control insect populations, thereby
decreasing the use of dangerous pesticides. Radioactive material is also
used in gauges that measure the thickness of eggshells to screen out
thin, breakable eggs before they are packaged in egg cartons. In
addition, many of our foods are packaged in polyethylene shrink-wrap
that has been irradiated so that it can be heated above its usual
melting point and wrapped around the foods to provide an airtight
protective covering.
All
around us, we see reflective signs that have been treated with
radioactive tritium and phosphorescent paint. Ionizing smoke detectors,
using a tiny bit of americium-241, keep watch while we sleep. Gauges
containing radioisotopes measure the amount of air whipped into our ice
cream, while others prevent spillover as our soda bottles are carefully
filled at the factory.
Engineers
also use gauges containing radioactive substances to measure the
thickness of paper products, fluid levels in oil and chemical tanks, and
the moisture and density of soils and material at construction sites.
They also use an x-ray process, called radiography, to find otherwise
imperceptible defects in metallic castings and welds. Radiography is
also used to check the flow of oil in sealed engines and the rate and
way that various materials wear out. Well-logging devices use a
radioactive source and detection equipment to identify and record
formations deep within a bore hole (or well) for oil, gas, mineral,
groundwater, or geological exploration. Radioactive materials also power
our dreams of outer space, as they fuel our spacecraft and supply
electricity to satellites that are sent on missions to the outermost
regions of our solar system.
Nuclear Power Plants
Electricity
produced by nuclear fission — splitting the atom — is one of the
greatest uses of radiation. As our country becomes a nation of
electricity users, we need a reliable, abundant, clean, and affordable
source of electricity. We depend on it to give us light, to help us
groom and feed ourselves, to keep our homes and businesses running, and
to power the many machines we use. As a result, we use about one-third
of our energy resources to produce electricity.
Electricity
can be produced in many ways — using generators powered by the sun,
wind, water, coal, oil, gas, or nuclear fission. In America, nuclear
power plants are the second largest source of electricity (after
coal-fired plants) — producing approximately 21 percent of our Nation's
electricity.
The purpose of a nuclear power plant is to boil water to produce steam to power a generator to produce electricity.
While nuclear power plants have many similarities to other types of
plants that generate electricity, there are some significant
differences. With the exception of solar, wind, and hydroelectric
plants, power plants (including those that use nuclear fission) boil
water to produce steam that spins the propeller-like blades of a turbine
that turns the shaft of a generator. Inside the generator, coils of
wire and magnetic fields interact to create electricity. In these
plants, the energy needed to boil water into steam is produced either by
burning coal, oil, or gas (fossil fuels) in a furnace, or by splitting
atoms of uranium in a nuclear power plant. Nothing is burned or exploded
in a nuclear power plant. Rather, the uranium fuel generates heat
through a process called fission.
Nuclear
power plants are fueled by uranium, which emits radioactive substances.
Most of these substances are trapped in uranium fuel pellets or in
sealed metal fuel rods. However, small amounts of these radioactive
substances (mostly gases) become mixed with the water that is used to
cool the reactor. Other impurities in the water are also made
radioactive as they pass through the reactor. The water that passes
through a reactor is processed and filtered to remove these radioactive
impurities before being returned to the environment. Nonetheless, minute
quantities of radioactive gases and liquids are ultimately released to
the environment under controlled and monitored conditions
The
U.S. Nuclear Regulatory Commission (NRC) has established limits for the
release of radioactivity from nuclear power plants. Although the
effects of very low levels of radiation are difficult to detect, the
NRC's limits are based on the assumption that the public's exposure to
man-made sources of radiation should be only a small fraction of the
exposure that people receive from natural background sources.
Experience
has shown that, during normal operations, nuclear power plants
typically release only a small fraction of the radiation allowed by the
NRC's established limits. In fact, a person who spends a full
year at the boundary of a nuclear power plant site would receive an
additional radiation exposure of less than 1 percent of the radiation
that everyone receives from natural background sources. This
additional exposure, totaling about 1 millirem (a unit used in measuring
radiation absorption and its effects), has not been shown to cause any
harm to human beings.
In agriculture
Radioisotopes
are used to induce mutations in plants in order to develop superior
varieties that are harder and more resistant to diseases.
Artificial Radioactivity
Difference between Natural and Artificial Radioactivity
Distinguish between natural and artificial radioactivity
Artificial radioactivity is the phenomenon by which even light elements are made radioactive by artificial or induced methods.
Artificial radioactivity
occurs when a previously stable material has been made radioactive by
exposure to specific radiation. Most radioactivity does not induce other
material to become radioactive. This Induced radioactivity was
discovered by Irène Curie and F. Joliot in 1934. This is also known as
man-made radioactivity. The phenomenon by which even light elements are
made radioactive by artificial or induced methods is called artificial
radioactivity.
Curie
and Joliot showed that when lighter elements such as boron and
aluminium were bombarded with α-particles, there was a continuous
emission of radioactive radiations, even after the α−source had been
removed. They showed that the radiation was due to the emission of a
particle carrying one unit positive charge with mass equal to that of an
electron.
Neutron
activation is the main form of induced radioactivity, which happens
when free neutrons are captured by nuclei. This new heavier isotope can
be stable or unstable (radioactive) depending on the chemical element
involved.
Because
free neutrons disintegrate within minutes outside of an atomic nucleus,
neutron radiation can be obtained only from nuclear disintegrations,
nuclear reactions, and high-energy reactions (such as in cosmic
radiation showers or particle accelerator collisions). Neutrons that
have been slowed down through a neutron moderator (thermal neutrons) are
more likely to be captured by nuclei than fast neutrons.
Methods of Producing Artificial Radioactive Isotopes
Describe methods of producing artificial radioactive isotopes
Methods of inducing radioactivity
- Nuclear activation:Neutron activation is the process in which neutron radiation induces radioactivity in materials, and occurs when atomic nuclei capture free neutrons, becoming heavier and entering excited states. The excited nucleus often decays immediately by emitting gamma rays, or particles such as beta particles, alpha particles, fission products and neutrons (in nuclear fission). Thus, the process of neutron capture, even after any intermediate decay, often results in the formation of an unstable activation product. Such radioactive nuclei can exhibit half-lives ranging from small fractions of a second to many years.
- Photonuclear reactions: A photonuclear reaction is a reaction resulting from an interaction between a photon and a nucleus.-During a photonuclear reaction energy of a gamma-ray photon is fully or partially absorbed by the nucleus forcing it into and excited state. From this excited state the nucleus can emit any particle, provided it has enough energy for such a process to occur. Most commonly it will emit a photon, but also a neutron (n), a proton (p) or an alpha (α) particle can be emitted.
Applications of Artificial Radioactivity
Mention the applications of artificial radioactivity
Application of artificial radioactivity include:
- Radiation safety:For physicians and radiation safety officers, activation of sodium in the human body to sodium-24, and phosphorus to phosphorus-32, can give a good immediate estimate of acute accidental neutron exposure.
- Neutron detection:One way to demonstrate that nuclear fusion has occurred inside a fusor device is to use a Geiger counter to measure the gamma ray radioactivity that is produced from a sheet of aluminum foil.In the ICF fusion approach, the fusion yield of the experiment (directly proportional to neutron production) is usually determined by measuring the gamma-ray emissions of aluminum or copper neutron activation targets.Aluminum can capture a neutron and generate radioactive sodium-24, which has a half-life of 15 hours[7][8] and a beta decay energy of 5.514 MeV.The activation of a number of test target elements such as sulfur, copper, tantalum and gold have been used to determine the yield of both pure fissionand thermonuclearweapons.
- Materials analysis:Main article: neutron activation analysis. Neutron activation analysis is one of the most sensitive and accurate methods of trace element analysis. It requires no sample preparation or solubilization and can therefore be applied to objects that need to be kept intact such as a valuable piece of art. Although the activation induces radioactivity in the object, its level is typically low and its lifetime may be short, so that its effects soon disappear. In this sense, neutron activation is a non-destructive analysis method.
- The potential use of photonuclear reactions for a range of applications is described. These are: photonuclear transmutation doping of semiconductors, neutron production from electron linacs, quality checking of radioactive waste, fission product incineration, photoexcitation of isomers for dosimetry, and nuclear resonance fluorescence for materials analysis. Initial brief descriptions of atomic and nuclear interactions of photons and of bremsstrahlung are given.
Radiation Hazards and Safety
The Effects of Nuclear Radiation on Human Body
Explain the effects of nuclear radiation on human body
Certain
body parts are more specifically affected by exposure to different
types of radiation sources. Several factors are involved in determining
the potential health effects of exposure to radiation. These include:
- The size of the dose (amount of energy deposited in the body)
- The ability of the radiation to harm human tissue
- Which organs are affected
The
most important factor is the amount of the dose - the amount of energy
actually deposited in your body. The more energy absorbed by cells, the
greater the biological damage. Health physicists refer to the amount of
energy absorbed by the body as the radiation dose. The absorbed dose,
the amount of energy absorbed per gram of body tissue, is usually
measured in units called rads. Another unit of radation is the rem, or
roentgen equivalent in man. To convert rads to rems, the number of rads
is multiplied by a number that reflects the potential for damage caused
by a type of radiation. For beta, gamma and X-ray radiation, this number
is generally one. For some neutrons, protons, or alpha particles, the
number is twenty.
- Hair:The losing of hair quickly and in clumps occurs with radiation exposure at 200 rems or higher.
- Brain:Since brain cells do not reproduce, they won't be damaged directly unless the exposure is 5,000 rems or greater. Like the heart, radiation kills nerve cells and small blood vessels, and can cause seizures and immediate death.
- Thyroid:The certain body parts are more specifically affected by exposure to different types of radiation sources. The thyroid gland is susceptible to radioactive iodine. In sufficient amounts, radioactive iodine can destroy all or part of the thyroid. By taking potassium iodide can reduce the effects of exposure.
- Blood System:When a person is exposed to around 100 rems, the blood's lymphocyte cell count will be reduced, leaving the victim more susceptible to infection. This is often refered to as mild radiation sickness. Early symptoms of radiation sickness mimic those of flu and may go unnoticed unless a blood count is done. According to data from Hiroshima and Nagaski, show that symptoms may persist for up to 10 years and may also have an increased long-term risk for leukemia and lymphoma. For more information, visit Radiation Effects Research Foundation.
- Heart:Intense exposure to radioactive material at 1,000 to 5,000 rems would do immediate damage to small blood vessels and probably cause heart failure and death directly.
- Gastrointestinal Tract:Radiation damage to the intestinal tract lining will cause nausea, bloody vomiting and diarrhea. This is occurs when the victim's exposure is 200 rems or more. The radiation will begin to destroy the cells in the body that divide rapidly. These including blood, GI tract, reproductive and hair cells, and harms their DNA and RNA of surviving cells.
- Reproductive Tract:Because reproductive tract cells divide rapidly, these areas of the body can be damaged at rem levels as low as 200. Long-term, some radiation sickness victims will become sterile.
Radiation sickness
Radiation
sickness results when humans (or other animals) are exposed to very
large doses of ionizing radiation. Radiation exposure can occur as a
single large exposure (acute), or a series of small exposures spread
over time (chronic). Exposure may be accidental or intentional (as in
radiation therapy).
Causes
- Accidental exposure to high doses of radiation such as a nuclear power plant accidents.
- Exposure to excessive radiation for medical treatments.
Symptoms
- Bleeding from the nose, mouth, gums, and rectum
- Bloody stool
- Bruising
- Confusion
- Dehydration
- Diarrhea
- Fainting
- Fatigue
- Fever
- Hair loss
- Inflammation of exposed areas (redness, tenderness, swelling, bleeding)
- Mouth ulcers
- Nausea and vomiting
- Open sores on the skin
- Skin burns (redness, blistering)
- Sloughing of skin
- Ulcers in the esophagus, stomach or intestines
- Vomiting blood
- Weakness
First Aid
- Check the person's breathing and pulse.
- Start CPR, if necessary.
- Remove the person's clothing and place the items in a sealed container. This stops ongoing contamination.
- Vigorously wash body with soap and water.
- Dry the body and wrap with soft, clean blanket.
- Call for emergency medical help or take the person to nearest emergency medical facility if you can do so safely.
How to Protect yourself from Nuclear Radiation Hazards
Protect himself/herself from nuclear radiation hazards
Precautions
- Time:An average the procedure time for a diagnostic coronary angiogram is approximately 30 minutes and an interventional procedure PCI or EPS/pacing would take between 90 to 120 minutes. However the fluoroscopic and the cine screening time are highly variable depending on the nature of the procedure and the experience of the operator. The lower the amount of time spent in a radiation area, the lower the exposure will be. Significant reductions can be achieved when an activity is delayed until after cine imaging is completed. Every effort should be made by the operating cardiologist in the cath lab to minimise fluoroscopy and cine screening time.
- Distance:Increasing the distance from the radiation beam decreases the risk of exposure. doubling the distance between the primary beam and operator, reduces the exposure by a factor of four. In addition, the radiation exposure varies according to the angle at which the camera is projected Oblique views (left and right anterior oblique) and steep angulations increase radiation exposure but are often employed to improve visualisation. 60-degree angulations give up to three times the operator dose than 30-degree angulations (11). The second operator or assistant is generally less exposed to radiation compared to the first operator but certainly more at risk than the other staff in the room.
- Shielding:Lead shields and shielding will significantly reduce the risk of exposure but only if appropriately used and in proper working order. Protective equipment includes lead aprons, thyroid collars and leaded glasses. With the newly designed frames and ultra light lenses, protective leaded eyewear is now used by more of the cardiologists and staff in cardiac cath lab. Some cath labs also use overhanging lead screens to prevent radiation exposure to brain. The staff should wear a protective apron of at least 0.25 mm lead equivalent. Protective gloves should be of at least 0.35 mm lead equivalent. All such protective clothing should bear an identifying mark and should be examined at yearly intervals. Defective items should be withdrawn from use.
- Adhering to guideline and protocols:Every unit or work place that deals with ionising radiation should have their own local guidelines and rules for radiation safety. These must be read, understood and strictly adhered to in daily practice. Staff must comply with these local rules in order to insure that the Trust and all their employees do not contravene statutory requirements of the ionising radiation regulations and other relevant legislation.
- Minimising risk of exposure to staff and patients: The occupational limit of radiation exposure in the UK currently is estimated at 20 mSv per year averaged over five consecutive years (5). Every operator who undertakes a cardiovascular procedure in the cath lab is responsible for the amount of radiation exposure to the patient, his or her co-staff and to themselves. In the event of an incident where the patient might have been exposed to inadvertent excess radiation either due to clinical circumstances, malfunctioning of the equipment or operation errors, the radiation protection adviser should be informed of the incident. It is their duty to estimate the radiation dose received by the patient and also advise whether the incident is to be reported.Only essential staff shall be in the cath lab during radiation exposure. All persons not required in the room should leave the room during serial radiographic exposure. The operator shall stand behind a barrier if possible. People who must move around the room during the procedure should wear a wraparound protective garment. When possible, the cardiologist and all other personnel required in the room should step back from the table and behind portable shields during cine and serial radiography procedures. This action can decrease the exposure of the cardiologist and the other nearby personnel by a factor of three or more (10).
Nuclear Fission and Fusion
The Nuclear Fission and Fusion
Explain the nuclear fission and fusion
Nuclear fission
Nuclear
fission is either a nuclear reaction or a radioactive decay process in
which the nucleus of an atom splits into smaller parts (lighter nuclei).
The
fission process often produces free neutrons and photons (in the form
of gamma rays), and releases a very large amount of energy even by the
energetic standards of radioactive decay. It is an exothermic reaction
which can release large amounts of energy both as electromagnetic
radiation and as kinetic energy of the fragments (heating the bulk
material where fission takes place).
In
order for fission to produce energy, the total binding energy of the
resulting elements must be less negative (higher energy) than that of
the starting element.
Nuclear fusion
Nuclear
fusion is a nuclear reaction in which two or more atomic nuclei come
very close and then collide at a very high speed and join to form a new
type of atomic nucleus.
During this process, matter is not conserved because some of the matter of the fusing nuclei is converted to photons (energy).
The
fusion of two nuclei with lower masses than iron (which, along with
nickel, has the largest binding energy per nucleon) generally releases
energy, while the fusion of nuclei heavier than iron absorbs energy.
The
opposite is true for the reverse process, nuclear fission. This means
that fusion generally occurs for lighter elements only, and likewise,
that fission normally occurs only for heavier elements.
Application of Nuclear Fission and Fusion
Mention the applications of nuclear fission and fusion
Nuclear fission is used in:
- Nuclear power plants to generate electricity for domestic and industrial use.
- In making nuclear bombs.
Nuclear fusion is used in:
- In fussion power plants to make electricity.
- To make nuclear weapons such as the hydrogen bombs.
Thermionic Emission
Thermionic
emission is the discharge of electrons from heated materials, widely
used as a source of electrons in conventional electron tubes (e.g.,
television picture tubes) in the fields of electronics and
communications. The phenomenon was first observed (1883) by Thomas A.
Edison as a passage of electricity from a filament to a plate of metal
inside an incandescent lamp. The classical example of thermionic
emission is the emission of electrons from a hot cathode into a vacuum
(also known as thermal electron emission or the Edison effect) in a
vacuum tube. The hot cathode can be a metal filament, a coated metal
filament, or a separate structure of metal or carbides or borides of
transition metals. Vacuum emission from metals tends to become
significant only for temperatures over 1000 K. The science dealing with
this phenomenon has been known as "thermionics," but this name seems to
be gradually falling into disuse.
Cathode Rays
Cathode
rays (also called an electron beam or e-beam) are streams of electrons
observed in vacuum tubes.Electrons were first discovered as the
constituents of cathode rays. In 1897 British physicist J. J. Thomson
showed the rays were composed of a previously unknown negatively charged
particle, which was later named the electron. Cathode ray tubes (CRTs)
use a focused beam of electrons deflected by electric or magnetic fields
to create the image in a classic television set.
The Production of Cathode Rays
Explain the production of cathode rays
Cathode
rays are so named because they are emitted by the negative electrode,
or cathode, in a vacuum tube. To release electrons into the tube, they
first must be detached from the atoms of the cathode.
Modern
vacuum tubes use thermionic emission, in which the cathode is made of a
thin wire filament which is heated by a separate electric current
passing through it. The increased random heat motion of the filament
atoms knocks electrons out of the atoms at the surface of the filament,
into the evacuated space of the tube.
Since
the electrons have a negative charge, they are repelled by the cathode
and attracted to the anode. They travel in straight lines through the
empty tube. The voltage applied between the electrodes accelerates these
low mass particles to high velocities. Cathode rays are invisible, but
their presence was first detected in early vacuum tubes when they struck
the glass wall of the tube, exciting the atoms of the glass and causing
them to emit light, a glow called fluorescence.
Researchers
noticed that objects placed in the tube in front of the cathode could
cast a shadow on the glowing wall, and realized that something must be
travelling in straight lines from the cathode.
After
the electrons reach the anode, they travel through the anode wire to
the power supply and back to the cathode, so cathode rays carry electric
current through the tube. The current in a beam of cathode rays through
a tube can be controlled by passing it through a metal screen of wires
(a grid) to which a small voltage is applied.
The
electric field of the wires deflects some of the electrons, preventing
them from reaching the anode. Thus a small voltage on the grid can be
made to control a much larger voltage on the anode. This is the
principle used in vacuum tubes to amplify electrical signals.
High
speed beams of cathode rays can also be steered and manipulated by
electric fields created by additional metal plates in the tube to which
voltage is applied, or magnetic fields created by coils of wire
(electromagnets). These are used in cathode ray tubes, found in
televisions and computer monitors, and in electron microscopes.

The Properties of Cathode Rays
State the properties of cathode rays
Properties of Cathode Rays Include:
- Cathode rays travel in straight lines. That is why, cathode rays cast shadow of any solid object placed in their path. The path cathode rays travel is not affected by the position of the anode.
- Cathode rays consist of matter particles, and posses energy by the virtue of its mass and velocity. Cathode rays set a paddle wheel into motion when it is placed in the path of these rays one the bladder of the paddle wheel.
- Cathode rays consist of negatively charged particles. When cathode rays are subjected to an electrical field, these get deflected towards the positively charge plate (Anode).We know that a positively charged body would attract only a negatively charged body, therefore the particles of cathode rays carry negative charge.Cathode rays also get deflected when these are subjected to a strong magnetic field.
- Cathode rays heat the object only which they fall. The cathode ray particles possess kinetic energy. When these particles strike an object, a part of the kinetic energy is transferred to the object. The causes a rise in the temperature of the object.
- Cathode rays cause green fluorescence on glass surface, i.e., the glass surface only which the cathode rays strike show a colored shine.
- Cathode rays can penetrate through thin metallic sheets.
- Cathode rays ionize the gases through which they travel.
- Cathode rays when fall only certain metals such as copper, but rays produced. The X-rays are not deflected by electrical or magnetic fields. X-rays pass through opaque materials such as black paper, but stopped by solid objects such as bones.
- Cathode rays travel with speed nearly equal to that of light.
The Application of Cathode Ray Tube
State the application of cathode ray tube
Application of cathode ray tube includes:
Televisions
Before
LCD or Plasma television, the CRT was used to create a moving image.It
used the same principle as a CRT, and for Black and White televisions,
that worked fine. B&W TVs were essentially the same thing as a CRT,
as all that's needed is the control of the brightness of the beam.
A
CRT TV works by having the electron beam "scan" the screen at an rate
faster than our eyes can perceive.This means that it shoots across the
screen like a machine gun, and the images we see are actually made from
many fluorescent dots.
The
fluorescence caused by the beam striking the screen lasts a bit longer
so that the next scan can be made without the previous image
disappearing.It scans twice each time, first filling in the odd "holes"
then the even ones.Each scan is about 1/50 of a second.
Colour
CRT TVs had 3 electron guns rather than a single one, a shadow mask,
and a modified fluorescent screen.The 3 electron guns were needed as
there were three primary colours (Red, Green and Blue) that could be
adjusted in different amounts to create any colour.
The
colours are formed as a result of the shadow mask, which is a layer
with holes in it that controls the angle of the incoming electron beams.
This is because the fluorescent screen is separated into multi-coloured
phosphors that are placed adjacent to each other at small intervals.
Thus it isn't actually a single coloured pixel, but rather 3 very small pixels that join together to form a larger dot.
Cathode Ray Oscilloscopes
A
Cathode Ray Oscilloscope (CRO) is a diagnostic device that allows one
to "see" voltage.It is essential a Cathode Ray Tube with two
perpendicular sets of deflecting electric plates.The vertical set is
where an input voltage is plugged in for the oscilloscope to display.
However,
the horizontal set is connected to a "sweep generator".This is what
provides a constant, but adjustable, timebase for the sweeping.It
essentially creates a "sawtooth voltage."This is what causes the image
to be animated, and measured with a linear scale.
X-Rays
The Structure and Mode of Action of the X-ray Tube
Describe the structure and mode of action of the x-ray tube
X-radiation
(composed of X-rays) is a form of electromagnetic radiation.
Electromagnetic radiation (EM radiation or EMR) is a form of radiant
energy released by certain electromagnetic processes. Visible light is
one type of electromagnetic radiation, other familiar forms are
invisible electromagnetic radiations such as X-rays and radio waves.
Most
X-rays have a wavelength ranging from 0.01 to 10 nanometers,
corresponding to frequencies in the range 30 pentahertz to 30 exahertz
(3×1016 Hz to 3×1019 Hz) and energies in the range 100 eV to 100
keV.X-ray wavelengths are shorter than those of UV rays and typically
longer than those of gamma rays.
In
many languages, X-radiation is referred to with terms meaning Röntgen
radiation, after Wilhelm Röntgen, who is usually credited as its
discoverer, and who had named it X-radiation to signify an unknown type
of radiation.
X-ray tube
The
x-ray tube consists of an emitter (either a filament or a
cathode),which emits electrons into a vacuum and an anode to accelerate
the electrons. This establishes a flow of electrons through the tube.
These
electrons are reffered to as a beam. The cathode is in the form of a
filament which emits electrons when heated. The anode is made of copper
and also carries the target.
A
high p.d between the anode and the cathode is maintained by an external
high-voltage source. A battery that supplies high current is used to
heat the cathode filament, which in many cases is made of tungsten. The
cathode is in the form of a coil to provide high resistance to the
passing current.
Production of X-rays
The
electrons from the filament experiences the p. d and accelerated
towards the anode. When they hit the anode, they are stopped and thereby
transfer their energy to the electrons of the anode material. This
gives rise to x-rays.
It is only a very small percentage of their energy that is converted to x-rays, with the rest of it being transformed to heat.
Defference between Soft and hard X-rays and their Production
Distinguish between soft and hard x-rays and their production
X-rays may be classified as hard or soft depending on their wavelengths, which give rise to different properties.
Differences between hard and soft x-rays
| Hard x-rays | Soft x-rays |
| They have shorter wavelength(high frequency) | They have longer wavelength |
| They have higher energy | Have less energy |
| Thigher penetrating power | Lower penetrating power |
| Are produced by higher accelerating potential | Produced by lower accelerating potential |
| Have higher velocity | Have lower velocity |
The Properties of X-rays
State the properties of x-rays
Properties of x-rays include:
- They travel in straight lines.
- They readily penetrate matter.
- They are not affected by electric or magnetic fields(they have no charge).
- They cause fluorescence in certain substances.
- They can be detected by photographic emulsion.
- They ionise gases causing the gases to conduct electricity.
The Application of X-Rays in Daily Life
Identify the applications of x-rays in daily life
The following are some uses of x-rays:
- In the medical field
- Crystallography
- Astronomy
- X-ray microscopic analysis
- X-ray fluorescence
- Security installations
- Industries
FORM FOUR PHYSICS STUDY NOTES TOPIC 1: & TOPIC 2:
FORM FOUR PHYSICS STUDY NOTES TOPIC 3: & TOPIC 4:
FORM FOUR PHYSICS STUDY NOTES TOPIC 5: & TOPIC 6:
FORM FOUR PHYSICS STUDY NOTES TOPIC 7: GEOPHYSICS
O'LEVEL PHYSICS
PHYSICS FORM FOUR
PHYSICS FORM THREE
PHYSICS FORM TWO
PHYSICS FORM ONE









No comments