Home
/
CHEMISTRY
/
CHEMISTRY FORM THREE STUDY NOTES TOPIC 7: CHEMICAL KINETICS, EQUILIBRIUM AND ENERGETICS & TOPIC 8: EXTRACTIONS OF METALS
CHEMISTRY FORM THREE STUDY NOTES TOPIC 7: CHEMICAL KINETICS, EQUILIBRIUM AND ENERGETICS & TOPIC 8: EXTRACTIONS OF METALS
TOPIC 7: CHEMICAL KINETICS, EQUILIBRIUM AND ENERGETICS
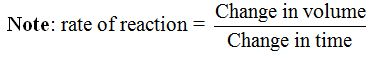
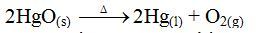
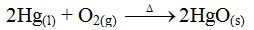
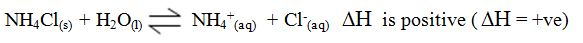
TOPIC 8: EXTRACTIONS OF METALS

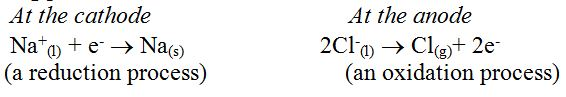 Chlorine is a valuable by-product of the decomposition process
Chlorine is a valuable by-product of the decomposition process
CHEMISTRY FORM THREE ALL TOPICS
CHEMISTRY FORM THREE TOPIC 1 & 2.
CHEMISTRY FORM THREE TOPIC 3 & 4.
CHEMISTRY FORM THREE TOPIC 5 & 6.
CHEMISTRY FORM THREE TOPIC 7.
CHEMISTRY FORM THREE TOPIC 8.
O'LEVEL CHEMISTRY
CHEMISTRY STUDY NOTES, FORM FOUR.
CHEMISTRY STUDY NOTES, FORM THREE.
CHEMISTRY STUDY NOTES, FORM TWO.
CHEMISTRY STUDY NOTES FORM ONE.
The Rate of Chemical Reactions
Copmarison between the Rates of Chemical Reactions
Compare the rates of chemical reactions
Chemical
reactions take place at different rates. Some are fast whereas others
are very slow. Let us consider the following reactions:
- Addition of sodium metal to water: 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) The reaction takes place immediately and violently. It is therefore a fast reaction.
- The rusting of iron in the presence of air and water giving hydrated iron (III) oxide, F2O3.XH2O: This is an extremely slow reaction.
These
two reactions could be taken as representative examples of extremely
fast and extremely slow reactions, respectively.There are, however,
other reactions which proceed at rates intermediate between these two
extremes. Rates of some of these reactions can be measured.
The
rate of a chemical reaction can be measured in various ways. Let us
consider the reaction between zinc and sulphuric acid to produce zinc
sulphate and hydrogen gas:
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
When zinc is added to dilute sulphuric acid in a flask, they react together. The zinc slowly disappears and the gas (H2)
bubbles off. After sometime, the bubbles of a gas form less quickly.
The reaction is slowing down. Finally, no more bubbles appear. The
reaction is over, because all the acid has been used up. Some zinc
remains behind in a beaker.
In
this reaction both zinc and sulphuric acid get used up in the reaction.
At the same time, zinc sulphate and hydrogen form. The rate of this
reaction could be determined by measuring any of the following:
- the amount of zinc used up per unit of time;
- the amount of sulphuric acid used up per unit of time;
- the amount of zinc sulphate produced per unit of time; or
- the amount of hydrogen produced per unit of time.
In general, the rate of a chemical reaction is determined by measuring the amount of reactant used up per unit of time or the amount of product produced
per unit of time. Therefore, the rate of a chemical reaction simply
refers to the amount of reaction which occurs in a unit time.
Experiments to Measure the Rates of Chemical Reactions
Perform experiments to measure the rates of chemical reactions
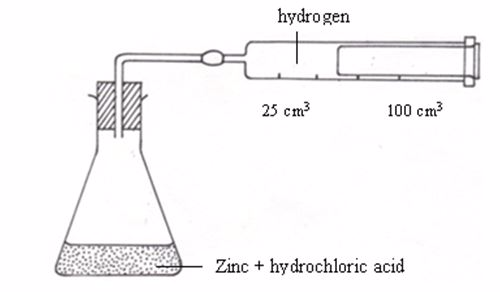
For
the reaction described above, it is easiest to measure the amount of
hydrogen produced per minute. The hydrogen can be collected as it
bubbles off and its volume can then be measured as shown in figure

Apparatus for measuring the production of gas
An
experiment may be designed to measure the volume of hydrogen produced
after every twenty seconds or so and then recording the data in a
notebook.The table below shows sample results from such an experiment.
| Time (s) | Volume of hydrogen gas (cm3) |
| 0 | 0 |
| 20 | 13 |
| 40 | 22 |
| 60 | 30 |
| 80 | 37 |
| 100 | 41 |
| 120 | 44 |
| 140 | 46 |
| 160 | 47 |
| 180 | 47 |
| 200 | 47 |
Questions from the experiment
Use these data to draw a graph of time (horizontal axis) against volume of hydrogen (vertical axis).
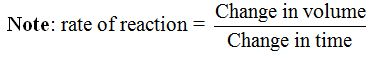
There are also other ways by which rates of chemical reactions can be measured. These include measuring the:
- change in intensity of colour:Many chemical reactions involve a change in colour. Potassium permanganate, for example, when it reacts with sulphur dioxide it changes from purple to colourless. The rate of such a reaction could be determined by measuring the rate at which the colour changes.
- formation or disappearance of a precipitate:The reaction between hydrochloric acid and sodium thiosulphate produce a yellow precipitate of sulphur. The rate at which this precipitate forms is a measure of the rate of a reaction.
Factors Affecting the Rate of Chemical Reactions
The Effect of Concentration on the Rate of a Reaction
Describe the effect of concentration on the rate of a reaction
A
chemical reaction will occur only if the particles of the reacting
substances are allowed to come in contact. Increasing the concentration
means increasing the density of the particles and hence the probability
of particles being close together and colliding more often. Thus, a
reaction can be made to go faster or slower by changing the
concentration of a reactant.
The
effect of concentration on the rate of reaction can be determined by
mixing dilute hydrochloric acid with sodium thiosulphate solution to
produce a precipitate of sulphur.Since this reaction produces a
precipitate from two different colourless solutions, the intensity of
the precipitate at any given moment in time represents the extent of the
reaction.
The experiment to determine this effect is carried out by mixing 2M hydrochloric solution acid with 50 cm3
of the thiosulphate solution (at different concentrations as shown in
the table below) and noting the time taken by the cross to disappear
using a stopclock. The procedure similar to that used to investigate the
effect of temperature above is used except that, in this particular
experiment, the concentration of the thiosulphate is altered each time
the experiment is repeated. The following table shows the results.
| Concentration of thiosulphate (g/dm3) | 10 | 20 | 30 | 40 | 50 |
| Time(s) for the cross to disappear | 250 | 120 | 65 | 32 | 15 |
As
the results show, a reaction goes faster when the concentration of the
thiosulphate (reactant) is increased. According to the above data, the
rate of the reaction approximately doubles as the concentration of the
thiosulphate is increased is increased by 10g dm-3.
The results clearly indicate that, as the concentration of the reaction is increased the time for the disappearance of the cross decreases, which chemically means that the rate of the reaction increases.
The Effect of Temperature on the Rate of a Reaction
Demonstrate the effect of temperature on the rate of a reaction
Increasing
the temperature of the system means increasing the kinetic energy and
hence the speed at which the reacting particles of the substance move.
Thus, the particles collide more often and combine to form new
substances. Temperature also provides the energy required to break the
bonds of a substance and hence enhances the decomposition or splitting
of a complex substance into more simpler substances. Therefore, an
increase in the temperature of the system will result to an increase in
the rate of reaction.
The
effect of temperature on the rate of reaction can be determined
experimentally. When dilute hydrochloric acid is mixed with sodium
thiosulphate solution, a fine yellow precipitate of sulphur forms.
2HCl(aq) + Na2S2O3(aq) → 2NaCl(aq) + SO2(g) + S(s) + H2O(l)
The solid sulphur (S(s))
produced in this reaction makes the colourless solution go cloudy. The
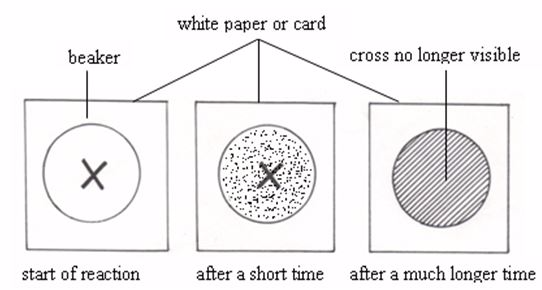
reaction is usually carried out in a flask placed on a piece of white
paper, with a black cross marked on it. An experiment is designed such
that each time a different temperature is used, while keeping the
concentrations of the reactants constant.
At
the beginning of the reaction, the cross can be seen easily. As the
reaction goes on, more and more sulphur is deposited, the flask becomes
more and more cloudy and the cross gradually gets harder to see. At
last, the cross can no longer be seen. It is fully covered by the
precipitate of sulphur. Time taken by the cross to disappear at a given
temperature indicates the rate of reaction at that temperature. The
quicker the disappearance of the cross, the faster is the reaction and
vice versa.
The reaction rate can be determined by the following procedure:
- 50 cm3 of a solution of sodium thiosulphate (containing 40g per litre of the solution) is measured and put into a 100 cm3 beaker.
- A wire gauze is placed on a tripod stand, then the beaker is put on the gauze, and the solution is gently warmed with a Bunsen burner flame.
- A thick black cross is marked on a white piece of paper or cardboard.
- When the temperature reaches a little above 200C, the beaker is taken quickly and placed on the cross.
- 10 cm3 of 2M hydrochloric acid solution is added quickly to the beaker and a clock is started at the same time. The temperature of the mixture is noted.
- When the cross is no longer visible, the clock is stopped.
- The experiment is repeated more times at temperatures of 300C, 400C, 500C, 600C, etc. Each time 10 cm3 of 2M hydrochloric acid and 50 cm3 of the thiosulphate solution are used.

Measuring the effect of temperature on the rate of reaction
The following table shows the specimen results obtained from such an experiment:
| Temperature (0C) | 20 | 30 | 40 | 50 | 60 |
| Time (s) for cross to disappear | 200 | 125 | 50 | 33 | 24 |
The
results show that the higher the temperature the faster the cross
disappears, which in turn, means that the rate of the reaction
increases.Therefore, the data clearly indicate that a reaction goes
faster when the temperature is raised. When the temperature is increased
by 100C, the rate approximately doubles.
The Effect of Surface Area of a Solid on the Rate of a Reaction
Show the effect of surface area of a solid on the rate of a reaction
In
many reactions, one of the reactants is a solid. The reaction between
hydrochloric acid and calcium carbonate (marble chips) is one example.
Carbon dioxide gas is produced.
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) + CO2(g)
One
of the ways by which the rate of such a reaction can be increased is by
reducing the particle size of the solid substance (marble chips). If
this substance is grinded to fine powder or to small pellets, the
surface area of the marble is increased. Therefore, more of the marble
is exposed to the acid for efficient reaction. This leads to increase in
the rate of reaction. Increased rate of reaction results to increased
production of carbon dioxide gas. Hence, the rate of reaction can be
determined by measuring the time taken to produce a given volume of
carbon dioxide by reacting equal masses of whole marble and crushed (or
powdered) marble, and then comparing the two results. The data obtained
is recorded in a table as shown below:
| Time (s) | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | |
| Volume (cm3) | Whole marble | 0 | 18 | 24 | 32 | 38 | 45 | 53 | 62 | 72 | 80 | 80 |
| Grinded marble | 0 | 34 | 52 | 67 | 74 | 76 | 80 | 80 | 80 | 80 | 80 | |
The
data above shows that it takes 90 seconds for whole marble chips to
react to completion while for powdered marble it takes only 60 seconds.
Also there is seen a small increase in volume of carbon dioxide gas per
unit of time in the case of ungrounded (whole) marble as compared to the
powdered sample. This absolutely proves that an increase in the surface
area of marble (a solid reactant) increases the rate of reaction and
hence the rate of production of carbon dioxide gas.
The Effect of Catalyst on the Rate of a Reaction
Demonstrate the effect of catalyst on the rate of a reaction
A
catalyst usually increases the rate of a reaction and this is called
positive catalysis. In general, a catalyst will function even if present
in very small amounts. Hydrogen peroxide is a clear, colourless liquid.
It can decompose to water and oxygen:
2H2O2(aq) → 2H2O(l) + O2(g)
The
rate of decomposition (reaction) of the peroxide can be increased
tremendously by adding a very little of manganese (IV) oxide in the
reaction vessel.

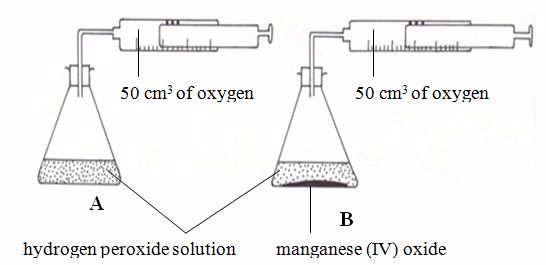
Investigating the effect of catalyst on rate of reaction
The reaction in flask A above is in fact very slow. It could take several days to produce just 50 cm3 of oxygen. In flask B only 1g of manganese (IV) oxide is added. The reaction goes very faster. 50 cm3 of oxygen is produced in a few minutes.
The
manganese (IV) oxide speeds up the reaction without being used up
itself. It is called a catalyst for the reaction.A catalyst is a substance that changes the rate of a chemical reaction but remains chemically unchanged at the end of the reaction.
Catalysts
for many reactions have been discovered. They are usually transition
metals or compounds of transition metals. There are also biological
catalysts, called enzymes. For example, the pancreatic juice secreted by
the pancreas contains enzymes that speed up digestion process.
Examples of catalysts for some common reactions are given in the following table:
| Reaction | Catalyst |
| Heating of potassium chlorate 2KClO3(s) → 2KCl(s) + 3O2(g) | Manganese (IV) oxide (MnO2) |
| Synthesis of sulphur trioxide 2SO2(g) + O2(g)⇔2SO3(g) | Vanadium (IV) oxide (V2O5) |
| Reduced iron powder |
| Manganese (IV) oxide or platinum powder |
Reversible and Irreversible Reactions
Comparison between Reversible and Irreversible Reactions
Compare reversible and irreversible reactions
Chemical reactions can be classified into two groups, that is, reversible and irreversible reactions.
Irreversible
reactions are those reactions which go to completion. In such
reactions, a known product is formed. The product(s) cannot be reversed
back to the original reactant(s). Consider the reaction between sodium
and chlorine to produce sodium chloride:2Na(s) + Cl2(g) → 2NaCl(s)
You
cannot turn sodium chloride back to chlorine gas and pure sodium metal
by ordinary means. This is a typical irreversible reaction.
Several
reactions are known to proceed in both directions, provided certain
conditions are maintained.A + B⇌ABSuch reactions are said to be
reversible. The sign ⇌indicates reversibility.
The Concept of Reversible and Irreversible Reactions
Describe the concept of reversible and irreversible reactions
A
reversible reaction is a chemical reaction in which the products can
react to re-form the reactants.In such reactions, reactants combine to
form products. However, under certain conditions, the products may be
converted back to reactants.The idea of “reactants” and “products” in
such circumstances is really confusing.
Example 1
Examples of reversible reactions
- When you heat blue crystals of copper (II) sulphate, they breakdown into anhydrous copper (II) sulphate, a white powder: CuSO4.5H2O(s)⇌CuSO4(s) + 5H2O(g)The reaction can be reversed by just adding water to the white powder, which quickly turns to blue crystals again. In fact, this is used as a test for water: CuSO4(s) + 5H2O(l)⇌CuSO4.5H2O(s)
- When you heat ammonium chloride (a solid) in the bottom of a test tube, it breaks down into ammonia and hydrogen chloride (gases). The gases readily combine at the top of the tube where it is cool: NH4Cl(s)⇌NH3(g) + HCl(g)This is a reversible reaction.
Equilibrium Reactions
Difference between Equilibrium Reactions and Simple Reversible Reactions
Differentiate equilibrium reactions from simple reversible reactions
Suppose
that 2 molecules of substance A reacts with 3 molecules of substance B
to produce 1 and 2 molecules of substances C and D respectively in a
homogenous system (i.e. entirely liquid or entirely gaseous).
2A + 3B⇌C + 2D
As
soon as a little of C and D are formed, a reverse reaction will begin.
At first the forward reaction will predominate, but, as C and D
accumulate the reverse reaction will build up until an equilibrium
position is reached, with forward and reverse reactions proceeding at
the same rate. The composition of the mixture will then appear constant,
though it is the net result of the two opposing reactions. Since
chemical equilibrium involves the balancing of two reactions which are
proceeding at the same time in opposite directions, it is said to be a dynamic equilibrium,
that is, it is an equilibrium involving the constant interchange of
particles in motion. Equilibrium is a dynamic condition in which two
opposing changes can occur at equal rates in a closed system. In a
closed system, matter cannot enter or leave, but energy can. Both matter
and energy can escape or enter an open system. For example, sunlight,
heat from a burner, or cooling by ice can cause energy to enter or leave
a system.
An
example of a real equilibrium reaction is the decomposition of mercury
(II) oxide. Mercury (II) oxide decomposes when heated to produce mercury
and oxygen gas.
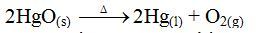
Again, mercury and oxygen combine to form mercury (II) oxide when heated gently
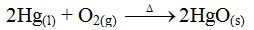
Suppose
mercury (II) oxide is heated in a closed system. Once the decomposition
has begun, the mercury and oxygen released can re-combine to form
mercury (II) oxide again. Thus, both reactions can proceed at the same
time. Under these conditions, the rate of the composition
(re-combination) reaction will eventually equal that of the
decomposition reaction. Then the reaction is said to be at equilibrium.
At equilibrium, mercury and oxygen will combine to form mercury (II)
oxide at the same rate that mercury (II) oxide decomposes into mercury
and oxygen. The amounts of mercury (II) oxide, mercury and oxygen can
then be expected to remain constant as long as these conditions persist.
At this point, a state of dynamic equilibrium has been reached between
the two chemical reactions. Both reactions continue, but there is no net
change in the composition of the system.
A
reversible reaction is in chemical equilibrium when the rate of its
forward reaction equals the rate of its reverse reaction, and the
concentrations of its reactants and products remain unchanged.
Two Equilibrium Reactions of Industrial Importance
Describe two equilibrium reactions of industrial importance
Conditions
for the industrial synthesis of different substances have to be
carefully chosen if the process is to be efficient and thus economically
viable. The considerations are:
- Yield – how much yield is produced?
- Rate – how fast is it produced?
- Energy – how much energy is lost during the process?
The
way in which this is achieved in practice is described briefly for
three important industrial reactions, namely, the Haber Process for the
manufacture of ammonia, the Contact Process for the manufacture of
sulphuric acid and the thermal dissociation of calcium carbonate for the
industrial manufacture of lime.
THE HABER PROCESS FOR THE INDUSTRIAL MANUFACTURE OF AMMONIA
The Haber process involves the reaction between nitrogen and hydrogen to produce ammonia:
N2(g) + 3H2(g)⇌2NH3(g)
A
German Chemist, Fritz Haber, was the first to show how this reaction
could be controlled to make useful amounts of ammonia. The first
industrial plant making ammonia by the Haber process opened in Germany
in 1913. Now over 100 million tones of ammonia are produced each year by
this process. Because of its importance, the Haber process for making
ammonia has been studied over a wide range of conditions of temperature
and pressure.
The effect of pressure
Ammonia
is produced from its elements by reduction of volume. Therefore, if the
system is in equilibrium and the pressure is raised, the equilibrium
will shift to the right. This is because the system will shift to favour
the side of the equation that has fewer molecules.
N2(g) + 3H2(g)⇌2NH3(g)
Therefore,
high pressure will increase the yield of ammonia. Modern industrial
plants use a pressure of 200-500 atmospheres. Pressure higher than this
range could be used, but high-pressure reaction vessels are expensive to
build.
The effect of temperature
The forward reaction producing ammonia is exothermic and the reverse reaction is therefore endothermic. Therefore, lowering the temperature will favour
ammonia production. However, the rate at which the ammonia is produced
will be so slow as to be uneconomical. So, it is necessary to include a
catalyst which will give sufficient reaction rate in spite of a
relatively low pressure. In practice, a compromise or optimum
temperature is used to produce enough ammonia at an acceptable rate.
Modern plants use temperatures of about 4500C.
The effect of concentration
If
the system is in equilibrium and more nitrogen is then added to
increase its concentration in the reaction mixture, the equilibrium
shifts to the right so as to tend to reduce the concentration of
nitrogen. That is, more ammonia will be produced to use up nitrogen.
Also, if hydrogen is added the equilibrium will, similarly, shift to the
right.
However,
in practice, there is no particular advantage in using excess of either
material (nitrogen or hydrogen) since the gases, nitrogen and hydrogen,
are mixed in a ratio of 3:1 by volume.Hydrogen is manufactured from
partial combustion of hydrocarbons, and nitrogen is obtained from the
air. They are mixed in the ratio of 3:1 proportion by volume and dried
(e.g. by silica gel). They are pre-heated by gases leaving the catalyst
chamber over the catalyst at 4500C. The ammonia produced is
absorbed in water or liquefied by refrigeration and the remaining
nitrogen and hydrogen gases are recycled.
If
the system was at equilibrium and then some of the ammonia was removed,
more ammonia would be produced to replace that which is removed. The
gas is removed from the reaction chamber when the percentage of ammonia
in the equilibrium mixture is 15%.
Based on the above discussions, the overall conditions used for the maximum production of ammonia are as follows:
- N2 and H2 are mixed in the ration of 3:1
- An optimum temperature of 4500C is chosen.
- A very high pressure (200-500 atm) is applied.
- A catalyst of finely divided reduced iron, usually promoted by alumina (aluminium oxide) is used.
- The ammonia is condensed and removed out of the reaction mixture and the remaining N2 and H2 recycled.
THE CONTACT PROCESS FOR THE INDUSTRIAL MANUFACTURE OF SULPHURIC ACID
The
first step in the production of sulphuric acid is the conversion of
sulphur dioxide to sulphur trioxide. The process involves the reaction
between sulphur dioxide and oxygen. The reaction is exothermic and
reversible.2SO2(g) + O2(g)⇌2SO3(g)
To
make sulphuric acid, the sulphur trioxide gas produced is dissolved in
98% concentrated sulphuric acid, forming “oleum” which is then diluted
with the correct amount of water to give ordinary concentrated sulphuric
acid.
- The effect of pressure:There are fewer gas molecules on the right of the equation. Therefore, increasing the pressure would favour the production of sulphur trioxide. In fact, the process is run at atmospheric pressure because the conversion of sulphur dioxide to sulphur trioxide is about 96% complete under these conditions. In practice, it is found that use of high pressure produces a very small gain in yield and involves extra cost. It is for this reason that ordinary atmospheric pressure is used.
- The effect of temperature:The reaction to produce sulphur trioxide is exothermic so, if the temperature is lowered the equilibrium shifts to the right. Hence, more sulphur trioxide will be produced. This means that sulphur trioxide production would be favoured by low temperatures. However, too low temperatures reduce the rate of reaction so increasing the time required for the production of the sulphur trioxide. This would mean an increase in production cost. For this reason, a catalyst must be introduced. A catalyst of vanadium (V) oxide is used to increase the rate of reaction. An optimum temperature of about 4500C is used. This gives sufficient sulphur trioxide at an economic rate. In general, low temperatures give an equilibrium favourable to an exothermic reaction, but catalysis is needed to give a favourable reaction rate.
- Effect of concentration of reactants:Suppose that in the contact process reaction, an equilibrium has been reached in certain conditions and oxygen is added to the system. According to Le Chatelier’s principle, the reaction would shift to the right so as to oppose this change, that is, to reduce the concentration of the oxygen added towards its former level. This can only be achieved by combining it with sulphur dioxide to form sulphur trioxide. So, increased concentration of oxygen favours conversion of more sulphur dioxide to trioxide. Likewise, a similar case can occur when sulphur dioxide is added to the system. In a similar manner, increased concentration of sulphur trioxide would favour the conversion of oxygen to sulphur trioxide and hence increased formation of the trioxide.In general, the conditions used in the contact process are as follows: An optimum temperature of about 4500C is chosen; A catalyst, vanadium (V) oxide, is used to speed up the reaction; An operating pressure of 1 atmosphere is applied. A higher pressure, which would theoretically increase the yield of SO3, is not used as it is uneconomical; Yield of SO3 would be increased by increasing either SO2 or O2. The cheaper reactant is O2.
Endothermic and Exothermic Reaction
The Concept of Endothermic and Exothermic Reactions
Explain the concept of endothermic and exothermic reactions
You
have met many different chemical reactions so far in chemistry. But
they all have one thing in common, that is, they involve an energy
change. The great majority of chemical reactions are accompanied by a
marked heat change.
During
chemical reactions as reactants form products, there is a change in
heat content. This is referred to as the enthalpy changeand is always
expressed in kilojoules per mole (kJmol-1). Two types of heat
change are distinguished. Those reactions that are accompanied by
evolution of heat to the surroundings are termed as exothermic reactions while those that are accompanied by absorption of heat from the surroundings are endothermic reactions.
- An exothermic reaction is one during which heat is liberated to the surroundings.
- An endothermic reaction is one during which heat is absorbed from the surroundings
When magnesium is burnt in air heat is evolved.
2Mg(s) + O2(g) → 2MgO(s)+ heat
The same case applies to the burning of coal in air.
C(s)+ O2(g) → CO2(g) + heat
Mixing sulphur nitrate and sodium chloride solutions gives a white precipitate of silver chloride and a temperature rise.
AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq)
When
ammonium nitrate is dissolved in water, there is a fall in temperature.
Also adding a mixture of citric acid and sodium bicarbonate to water
produces bubbles and a fall in temperature. In both reactions, the
temperature of the water falls because the reactions take heat energy
from it. These reactions are therefore endothermic.
The
heat changes that occur during any chemical reaction represent changes
in the energy content of the whole system. The energy content may
increase or decrease depending upon whether heat is absorbed or evolved.
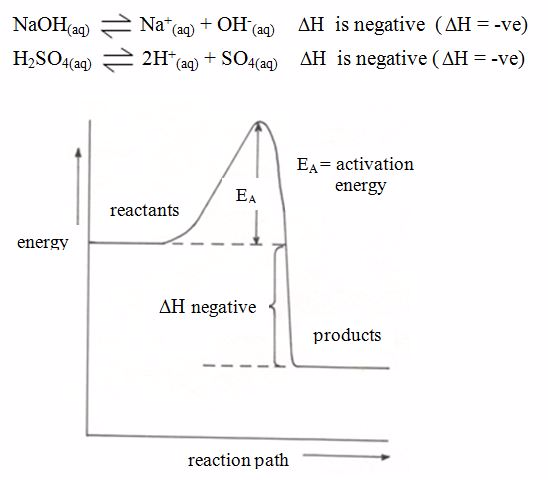
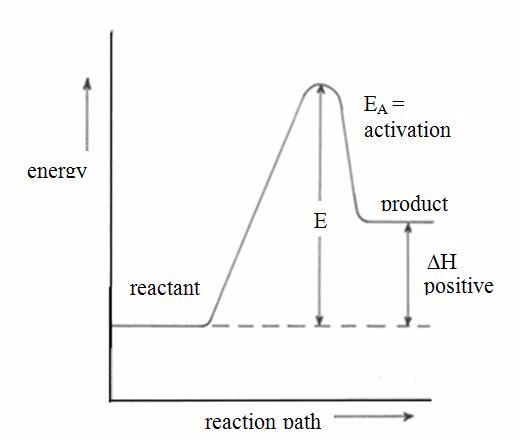
Energy Level Diagrams for Exothermic and Endothermic Reactions
Draw energy level diagrams for exothermic and endothermic reactions
For exothermic reactions,
the enthalpy changeis conventionally assigned a negative value. For
example, when pellets of sodium hydroxide or concentrated sulphuric acid
dissolve in water, heat is evolved and the system loses heat to the
surrounding.

Energy level diagram for exothermic reaction
For endothermic reactions,
the enthalpy changeis assigned a positive value. For example, when
potassium iodide or ammonium chloride dissolves in water, heat is
absorbed from the surroundings.
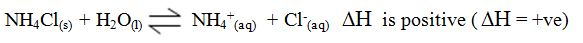

TOPIC 8: EXTRACTIONS OF METALS
Occurrence and Location of Metals in Tanzania
Locations of Important Metal Ores in Tanzania
Identify locations of important metal ores in Tanzania
Most metals are found naturally as compounds called minerals.
Rocks are made up of crystals of metals. An ore is a rock that contains
enough of a metal compounds for it to be worth extracting the metal.
The most common ores contain oxides. An example is the ore haematite,
which contains iron (III) oxide. Some contain other metal compounds.
Malachite contains copper (II) carbonate.
Tanzania
is blessed with an assortment of minerals. The mineral found in
Tanzania include gold, which is found in Geita (Mwanza), Kahama
(Shinyanga), Nyamongo, Buhemba and Majimoto (Mara), and Nzega (Tabora).
Large deposits of iron occur at Liganga in Ludewa (Iringa). Copper is
found in Mpanda (Rukwa). We also have sodium which occurs in the form of
sodium chloride, at Uvinza (Kigoma), in the form of soda ash (Na2CO3) in lake Natron, as a rock salt in Kilwa and as a dissolved salt (NaCl) in sea water.
The Abundances of Metals in the Earth’s Crust
Compare the abundances of metals in the earth’s crust
Metals
occur in the sea and in the earth. The form in which they occur depends
on their reactivity. The most reactive metals are extracted from the
sea. Calcium is extracted from limestone, chalk and marble in the sea.
Metals of medium reactivity are found in the earth in form of oxides and
sulphides. Examples of these metals include aluminium, zinc, iron and
tin. Least reactive metals such as silver and gold occur as free
uncombined elements.
We
obtain most of the metals we use from the earth’s crust. The majority
of metals are too reactive to exist as natural elements. They are found
as compounds in ores. The chief ores in order of their economic
importance are oxides, sulphides, chlorides and carbonates.
The
most common metal is aluminium, which makes about 7.8% of the earth’s
crust, followed by iron, which accounts to approximately 4.5%. Others
include calcium (3.5%), sodium (2.5%), potassium (2.5%) and magnesium
(2.8%). The remaining metals make about 3% of the earth’s crust.
Chemical Properties of Metals
The Difference between Physical and Chemical Strengths of Metals
Differentiate the physical and chemical strengths of metals
Metals are known to have a greater tendency to lose electrons in the course of which they go into solution as ions. A metal, therefore, may be defined as an element which can ionize by electron loss. The number of electrons lost per atom is the valency of the metal and the ion carries an equal number of positive charges, as:
- Na →Na+ + e- (univalent)
- Mg →Mg2+ + 2e- (divalent)
- Al →Al3+ + 3e- (trivalent)
The following are important chemical properties of metals:
- They react with oxygen to form oxides. For example, magnesium burns in air to form magnesium oxide. Metal oxides are bases, which mean they react with water to form an alkaline solution and with acids to form salts.
- Metals form positive ions when they ionize. Consider the ionization of sodium, magnesium and aluminium in the above equations in which case ionization resulted into Na+, Mg2+ , and Al3+ ions respectively. However, there are some exceptions. For example, hydrogen is a non-metal which forms positive ions in solution, H+. This is the only exception in this case.
Physical
strength is different from chemical strength. Physical strength is
tensile strength of the metal. There are metals with high tensile
strengths like iron, copper and aluminium. Other metals like sodium and
potassium have low tensile strengths..Chemical strength is the
reactivity of the metal. Sodium and potassium have very low tensile
strengths but they are the strongest metals chemically.
Sodium and potassium are very weak physically. Sodium is very soft metal and can be cut easily with a knife. It melts at 98oC.
It is so light that if floats on water, but it reacts immediately with
the water forming an alkaline solution. When freshly cut, it has a
silvery lustre but rapidly furnishes due to its reaction with
atmospheric carbon dioxide and moisture.
Potassium is a white metal with lustre. It melts at 63oC.
Like sodium, potassium is a very light metal and it can also float on
water, with which it reacts to form an alkaline solution.
However,
sodium and potassium are among the strongest metals chemically. These
metals are very reactive and they are always found combined with other
elements. The metals are so reactive that they will combine with any
non-metal nearby. They never occur free in nature. Both sodium and
potassium are so reactive that they have to be stored under oil to
prevent them coming into contact with water or air.The compounds of
sodium and potassium are quite abundant in nature.
The reactivity and tensile strengths of some metals
- Calcium:Calcium is amongst a group of metals that are too reactive to occur in the free state. It occurs mainly as carbonate, sulphate, fluoride and silicate. It is a soft, greyish metal. In comparison with potassium and sodium, it has a lower tensile strength and high density.
- Iron:Iron is a typical metal. Its density is 7.87. It melts at 1530oC. Iron is a moderately reactive metal. The metal reacts with excess steam at red heat to produce triiron tetraoxide.
- Copper Copper is a less reactive metal. It is a red-brown metal with a lustre. It can be polished. Its tensile strength is fairly high. When heated in air, copper forms a layer of black copper (II) oxide on the surface: 2Cu(s) + O2(g)→2CuO(s)It reacts with hot concentrated sulphuric acid to form copper (II) sulphate and liberate sulphur dioxide. Cu(s) + 2H2SO4(aq)→CuSO4(aq) + 2H2O(l) + SO2(g)
The table below compares the tensile strengths of calcium, iron and copper metals.
Tensile strengths, densities and melting points of calcium, iron and copper
| Calcium | Copper | Iron | |
| Tensile strength | Low | Fairly high | High |
| Melting point (oC) | 850 | 1080 | 1535 |
| Density (g cm-3) | 1.55 | 8.95 | 7.9 |
The Reducing Power of Different Metals
Compare the reducing Power of different metals
As we learned early, metals tend to lose electrons during chemical reactions. This process of losing electrons is called oxidation.
Metals normally lose electrons to non-metals, which accept those
electrons. Therefore, metals are said to be electron donors while
non-metals are electron acceptors. In this case, metals can be termed as
reducing agents, because they donate electrons which, when
accepted by non-metals, tend to lower their oxidation numbers.
Non-metals are called oxidizing agents, because they oxidize or increase the oxidation number of metallic atoms through accepting the electrons donated by metals.
Metals
differ in the ease of losing the electrons, depending on their
electronic configurations. This is because; it is only the outer
electron(s), which take part in a chemical reaction. The nucleus of an
atom, being positively charged, normally attracts the electrons towards
itself, hence making the electrons difficult to remove from their
shells. The further the electrons are from the central nucleus, the
easier it is removing them from their shells and vice versa. Therefore,
atoms with larger atomic radii donate their electrons more easily than
those with small atomic radii.
For
example, compare the electronic configuration of sodium (2:8:1) with
that of potassium (2:8:8:1). Sodium ionizes by losing one electron from
its outer shell to attain the structure of the nearest noble gas (2:8).
Likewise, potassium ionizes by losing one electron from its outer shell
to attain the noble gas structure, 2:8:8. But, in which of the two cases
is it easy to remove electron and why? It is easy to remove the
electron from the outer shell of potassium than sodium because the
atomic radius of calcium is too large as compared to that of sodium.
This is because in calcium the outer electron is more loosely held by
the central nucleus and hence easy to remove from its shell. This is
true because the ability of the nucleus to hold the electrons firmly
depends on the distance of the electrons from the nucleus.
Metals
that release the electrons more readily are said to be strong reducing
agents compared to those that release their electrons least readily. For
example, potassium with an electronic configuration of 2:8:8:1 is a
stronger reducing agent than sodium, which has an electronic
configuration of 2:8:1.
Therefore, it is clear that when atoms of elements lose electrons from their outermost shells, they obtain octet or doublet structures
and hence become chemically stable. It is not easy to remove
electron(s), by ordinary chemical means from such stable noble gas
structures.
From
the context of the above explanations, the reducing power of a metal
can be defined as its readiness to release electrons from its outer
shell. Metals whose atoms release electrons more readily have high
reducing powers than those metals whose atoms do not readily release
their electrons.
Reactivity series of metals
Reactivity
series refers to arranging or listing the metals in order of
reactivity. The reactivity series are obtained by consideration of the
action of air, water and acids on the metals,
and how easily the oxides of these metals can be reduced. Consider the
table of reactivity series below (Table 8.2). Oxides of the first group
of five metals cannot be reduced by carbon. Those of the second group of
three metals can react with acids, displacing hydrogen. The third and
last group comprises of least reactive metals. In table 8.2, the metals
are arranged in order of reactivity series. It indicates the inverse
order in which the elements were isolated. Thus, metals low in the
series such as gold, silver and lead have been known since early times.
Metals high in the series proved very difficult to isolate. It was
Davy’s work on electrolysis that led to isolation of potassium, sodium,
calcium, magnesium and aluminium over a period of years from 1807, when
Davy isolated potassium and sodium, to about 1850, when aluminium was
isolated.

The reactivity series of metals
Metals
low down in the series are frequently found as the free elements,
although they may also be obtained from ores because the amounts found
as the free metal are not sufficient for industrial purposes. Gold,
however, the last element of the series is found and mined almost
entirely as the free element.
Generally,
it is these relatively uncreative metals that we find the most uses
for. Iron and copper, for example, can be found in many household and
everyday objects.Metals higher up in the series are more reactive than
those lower down. A metal higher up in the series will displace a metal
lower down from a solution of one of its salts. For example, iron will
displace copper from its salt.
Fe(s) + CuSO4(aq)→ Cu(s) + Fe SO4(aq)
Zinc and iron will displace hydrogen from dilute acids such as dilute hydrochloric acid or dilute sulphuric acid.
Zn(s) + 2HCl(aq) →ZnCl2(aq) + H2(g)
Fe(s) + H2SO4(aq) →FeSO4(aq) + H2(g)
Copper
has no reaction on either dilute sulphuric acid or dilute hydrochloric
acid. With hot concentrated sulphuric acid, sulphur dioxide is liberated
and copper (II) sulphate is formed.
Cu(s) + 2H2SO4(l) →CuSO4(aq) + 2H2O(l) + SO2(g)
Useful things to remember about the reactivity series
- The more reactive the metal, the more compounds it forms. So only copper, silver and gold are ever found as free elements in the earth’s crust. The other metals are always found as compounds.
- When a metal reacts, it gives up electrons to form ions. The more reactive the metal, the more easily it gives up electrons.
- The more reactive the metal, the more stable its compounds are. Stable means difficult to breakdown. For example, when you heat sodium nitrate you get sodium nitrite:2NaNO3(s)→ 2NaNO2(s) + O2(g)But copper (II) nitrate breaks down further, to oxide, giving off nitrogen dioxide:2Cu(NO3)2(s)→ 2CuO(s) + 4NO2(g) + O2(g)
- The more reactive the metal, the more difficult it is to extract from its compounds (since the compounds are stable). For the most reactive metals, you will need the toughest method of extraction: electrolysis.
- The less reactive metals have been known and used since ancient times, because they are easiest to extract.6. If you stand two metals in an electrolyte and join them up with a copper wire, electrons will flow from the more reactive metal to the less reactive one.
Extraction of Metals by Electrolytic Reduction
The Criteria for the Choice of the Best Methods of Extracting a Metal from its Ore
Outline the criteria for the choice of the best methods of extracting a metal from its ore
Most metals are naturally found in compounds called ores. The metals are obtained from the ores by a process calledextraction.The extraction consists of separating the metal from minerals in the ore
A few metals are found “native”. This means that they are uncombined with other elements, for example, silver and gold.To
extract ores containing minerals, a chemical reaction must be used to
separate the metal from other elements. The choice of the best method
for extraction of a metal from its ore depends on the chemical
reactivity of the metal to be extracted. Most ores contain metal oxides.
To extract the metal, oxygen must be removed from it. This reaction if
called reduction.
Because
the metal is always the most electropositive part of an ore, and so has
a positive oxidation state, the formation of the free metal from its
ore is always a reduction.
Mn+ + ne- →Mo
Very
reactive metals like sodium and potassium are strongly bonded in their
ores. The more reactive the metal is the more stable its compounds are,
and the more energy is needed to break down the bond between the metal
and oxygen. Therefore, the extraction of these metals requires a strong
method of reduction: electrolysis (or electrolytic reduction).
Hence,
metals at the top of the reactivity series are obtained from their ores
by electrolysis (electrolytic reduction). Electrolysis is a more
expensive process than reduction with carbon or carbon monoxide.
However, it is the only economic way to obtain metals such as aluminium.
For less reactive metals at the middle of the reactivity series, the
oxygen can be removed by chemical reduction with carbon or carbon
monoxide. This method is used for extraction of metals such as zinc,
iron and copper as shown and discussed in table 8.3. Least reactive
metals such as copper, silver and gold may be found in uncombined state.
Methods of extraction of different metals from their ores
| Metal | Method of extraction from ore |
| Potassium | Electrolysis |
| Sodium | |
| Calcium | |
| Magnesium | |
| Aluminium | |
| Zinc | Chemical reduction with carbon or carbon monoxide |
| Iron | |
| Lead | |
| Copper | Roasting in air |
| Silver | Occur naturally as elements |
| Gold |
Stages of the extraction of moderately reactive metals
There are four main stages for the extraction of moderately reactive metals:
- Mining and concentration of the ore
- Roasting in air
- Reduction of oxides to metals
- Purifying the metal
Mining and concentration of the ore
Mining is just the first step. The ore must then be decomposed to give the metal. This is called extraction.The unwanted rock material in the ore is called gangue. The ore is treated to remove as much gangue as possible. For example:
- It is crushed and washed. In this case, the ore is broken down into small pieces, which are then grinded down to fine powder. Then it is either dropped into water, where the fragments containing the metal sink faster or jets of air are blown at it, where the lighter waste material is carried to one side.
- A method called froth flotation is used with sulphide ores (e.g. CuS or ZnS). The ore is powdered, fed into water tanks and made into slurry with water. Then “frothing” chemicals (a suitable oil) are added. Sulphides are attracted to these chemicals. When air is blown through the slurry, froth rises to the top of the tank carrying the metal sulphides with it. They are skimmed off and dried. The gangue sinks.
- Magnetic separation can be used. The iron ore can be separated from other material in the crushed ore by using electromagnet.
Roasting in air
For
the ores that occur as sulphides or carbonates of the metal, the
concentrated ore is heated (roasted) in air to convert the ore into an
oxide, for example:
- 2PbS(s) + 3O2(g)→ 2PbO(s) + 2SO2(g)
- 2ZnS(s) + 3O2(g)→ 2ZnO(s) + 2SO2(g)
- ZnCO3(s)→ ZnO(s) + CO2(g)
It
is usual to convert sulphides and carbonates into oxides before
reduction because oxides are more easily and efficiently reduced than
sulphides. The oxides resulting from roasting (heating) the sulphides or
carbonates in air are then reduced chemically with carbon or carbon
monoxide.
Most copper is extracted from copper pyrites, CuFeS2. The ore is roasted in a limited supply of air to produce to produce copper (I) sulphide:
2CuFeS2(s) + 4O2(g) → Cu2S(s) + 3SO2(g) + 2FeO(s)
The copper (I) sulphide is then reduced to copper by heating in air: Cu2S(s) + O2(g) → 2Cu(s) + SO2(g)
The
copper produced by this method is never pure. It must be refined
(purified) by electrolysis if it has to be used for electrical wiring.
Reduction of oxides to metals
This
is another important stage in the extraction of metal. Here, the
roasted ore must now be reduced to respective metals. Reduction occurs
at a very high temperature. The materials employed for reduction are
mainly carbon or carbon monoxide. Thus;
Fe2O3(s) + 3CO (g) →2Fe (l) + 3CO2(g)
ZnO(s) + C(s) →Zn(s) + CO(g)
Purification (Refining)
Very
often, the product of the reduction process is never pure. The product
has to be purified first before being put into use. Purification is
normally done through a number of ways, which include:
- Electrolysis: Electrolysis is used to produce a pure metal directly from its molten compounds. Examples of metals which are purified by electrolysis are copper and zinc. Copper produced in large scale is purified by electrolysis, a process often called copper refining.
- Oxidation: The molten crude metal is exposed to hot air in a furnace. The impurities in the crude metal are oxidized with oxygen from the air. They escape as vapour or form a scum over the molten metal, which is then removed by skimming. However, this method is used only when the impurities have a greater affinity for oxygen than the metal. The method is applied in the manufacture of steel from pig iron and in the purification of tin and lead.
- Distillation:In distillation, the crude metal is heated in a furnace until the pure metal evaporates, leaving behind the impurities. The vapour is then collected and condensed in a separate chamber. This method forms an integral part in the extraction of zinc, cadmium and mercury. A further distillation, usually in vacuum, gives a very pure product.
- Formation of carbonyls: very pure nickel and iron are made by forming their volatile carbonyls, which are then decomposed by heating.
- Zone refining: This recently developed method is used to produce silicon and germanium of extreme purity. In this method, a small high-frequency induction furnace is placed round one end of a long rod of the metal and a thin cross-section of the metal is melted. The furnace is then moved slowly along the rod. Pure crystals of the metal separate from the melting metal but impurities remain in the liquid and are carried along to the other end.
The Extraction of Sudium from its Ore
Explain the extraction of sodium from its ore
Alkali
metals are strong reducing agents and cannot be extracted by chemical
reduction of their oxides or other compounds. The only possible method
of their extraction is by electrolysis of their fused chlorides.Sodium
is extracted industrially by electrolysis of either fused sodium
hydroxide (Castner’s process) or fused sodium chloride (Down’s process),
in which sodium chloride is electrolysed in the molten condition.
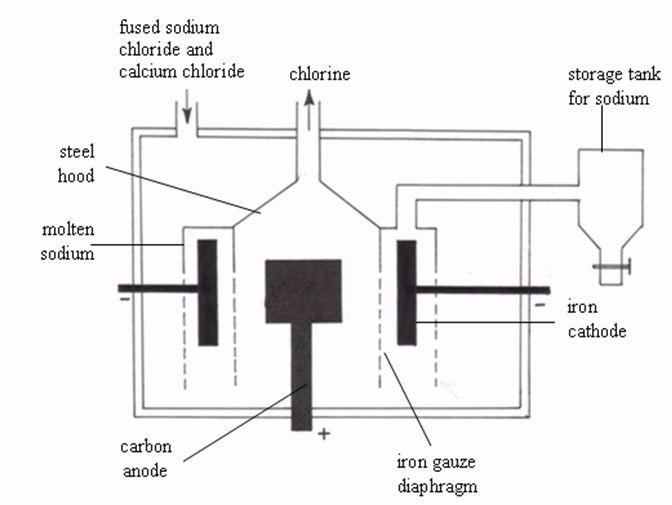
Electrolysis of sodium chloride by the down’s process
In
this case, fused sodium chloride is used. And because the melting point
of sodium chloride is high (about 800°C), calcium chloride is added to
lower the melting point to about 600°C and thus economize on electrical
power. The composition of the electrolyte is 40% sodium chloride and 60%
calcium chloride.The Down’s cell (figure 8.1) used for the extraction
of sodium consists of an iron box through the bottom of which rises a
circular carbon anode. A ring-shaped iron cathode surrounds this carbon
anode. The cathode is enclosed in iron gauze diaphragm, which also
separates the two electrodes. At 600°C, the sodium and chlorine produced
would react violently if allowed to come in contact. A diaphragm around
the anode, which keeps the two products apart, prevents this.
During
electrolysis, chlorine is librated at the anode and escapes via the
hood. Sodium is liberated at the cathode, collects in the inverted
trough placed over the cathode, rises up the pipe, and overflows into
the storage tank, from which it is tapped off through the iron vessel.
The
sodium metal is collected upwards in the Down’s cell because of its low
density which makes it float over the mixture. The sodium metal from
Down’s cell contains some calcium, which is also formed through
electrolysis. The calcium crystallizes when the mixture cools and a
relatively pure sodium metal is obtained.

The Down’s cell
Fused sodium chloride produces Na+ and Cl- ions, where Na+ moves to the cathode and Cl- moves to the anode. The reactions taking place at the electrodes are as follows:
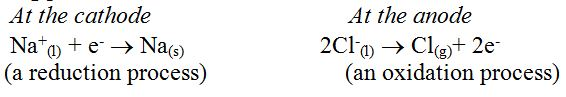
Chlorine gas is therefore formed at the anode while sodium metal is produced at the cathode.
2NaCl(l) →2Na(s) + Cl2(g)
Extraction of Metals by Chemical Reduction
The Extraction of Iron from its Ore
Describe the extraction of iron from its ore
Iron
is only second to aluminium as the most abundant metal in the earth’s
crust. Its chief ores are haematite, an impure iron (III) oxide, Fe2O3, which contains about 70% of iron; magnetite (or magnetic iron ore), triiron tetraoxide, Fe3O4, which contains 72.4% of iron; and spathic iron ore, iron (II) carbonate, FeCO3. It also occurs as limonite, Fe2O3.xH2O and as the sulphide in iron pyrites, FeS2.
However, though abundant in the earth’s crust, iron pyrite is not used a
source of iron. It is mainly used in the production of sulphuric acid.
To extract iron, three substances are needed:
- Iron ore: The chief ore is haematite. It is mainly iron (III) oxide, Fe2O3 mixed with sand.
- Limestone: This is mainly calcium carbonate, CaCO3.
- Coke: This is made from coal and is almost pure carbon.
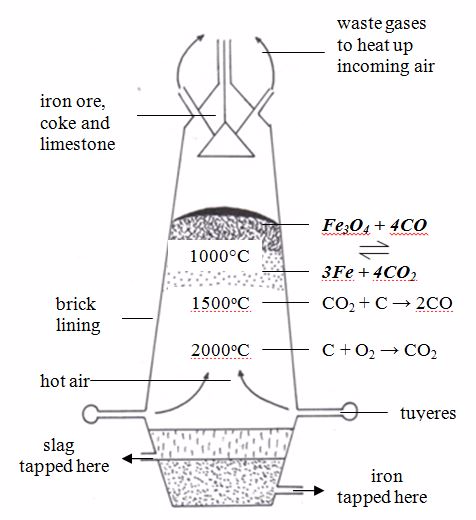
The extraction of iron in a blast furnace
Before being introduced into the blast furnace, all forms of iron ore must be converted to the oxide, Fe2O3, by roasting in air.
4FeCO3(s) + O2(g) →2Fe2O3(s) + 4CO2(g)
4FeS2(s) +11O2(g) →2Fe2O3(s) + 8SO2(g)
Iron ore, coke and limestone are mixed together to give a mixture called charge. The charge is introduced into a tall tower called a blast furnace
(figure 8.2). At the bottom of the furnace, hot air is blasted in
through several pipes known as tuyeres. A well at the bottom of the
furnace serves to hold the molten iron and slag until these can be run
off. The charge is fed in continuously from the top.
Reactions that occur in the furnace
- At the bottom of the furnace where temperature is the highest, air attacks the coke to produce carbon dioxide.C(s) + O2(g)→ CO2(g)
- In the middle of the furnace, the rising up carbon dioxide gas is reduced by more coke, producing carbon monoxide.C(s) + CO2(g)→ 2CO(g)
- At the top of the furnace, carbon monoxide reduces iron (III) oxide to metal. Fe2O3(s) + 3CO(g)→2Fe(s) + 3CO2(g)
The
molten iron trickles down the furnace and gathers at the bottom.
Periodically, this molten iron is tapped off and run into moulds (or
containers), where it is allowed to cool in long bars of about 1 metre
long and 10 cm in diameter. At this stage, it is called ‘cast iron’ or
‘pig iron’.
The hot waste gases, mainly nitrogen and oxides of carbon, escape from the top of the furnace and are used to heat incoming air. This helps to reduce the energy costs of the process.

The Blast furnace
Action of limestone
The
limestone, which is introduced together with the ore, is first
decomposed at this high temperature to form calcium oxide.CaCO3(s)→ CaO(s) + CO2(g)
The main impurity in iron ore is sand, made of silicon dioxide (SiO2). This reacts with calcium oxide to form calcium silicate.CaO(s) + SiO2(s) →CaSiO3(l)
The calcium silicate, which is in liquid state, falls to the bottom of the furnace. This liquid, called slag
(because it is less dense than the molten iron) is tapped off
separately. Slag is a useful by-product. It is used for making roads,
production of cement, and as a fertilizer.
Environmental Consideration
Environmental Destruction caused by Extraction of Metals
Identify environmental destruction caused by extraction of metals
Mining
and mineral extraction is important for economic development and
general human welfare. Without mining, we would have no cars, computers,
handsets, washing machines or other equipment that we use to simplify
our work and hence improve the quality of our lives. However, mining can
cause many environment problems. The following are some of the
environmental problems caused by mining:
- Land subsidence (sagging): Holes created due to underground mining cause land to sink (or subside). This is because the holes underneath the ground cause imbalance in weight of the soil above the ground. This may result to severe damage to buildings and other infrastructures such as roads, railway trucks and so forth.
- Poisonous substances: Poisonous compounds (for example of lead, cadmium and arsenic) are found in many ores. These may be washed into the soil and streams because of the mining process. If they happen to reach the water, they can kill fish and plant life, and can end up in your food as well. Gold extraction process makes use of mercury. If untreated effluent from the gold mine is directed to nearby rivers or streams, the metals may end up in fish, which might be someone’s food. Consumption of such fish can result to brain damage due to mercury contained in it.
- Large volume of waste: Large-scale mining operations inevitably produce a great deal of waste. This waste not only comprises of earth from the soil and gangue but also includes the toxic chemicals added to the ore to aid metal extraction. The waste material gets washed into streams and rivers. The sediment that builds up blocks rivers and alters their routes. This serves as a source of pollutants to natural water systems.
- Noise and dust: Mining activities produce a lot of noise and dust. Noise and dust can be caused by haulage trucks, rock blasting and crushing, drilling operations and heavy traffic. Everything for miles around the mine may get covered with dust.
- Big holes in the ground: Mineral extraction leads to boring of deep holes through the ground in the course of searching for rich ores. Huge amounts of rock are dug up to get a small amount of ore. For example, 1000 tonnes of rock may produce just 5 tonnes of copper. This leaves huge scars on the landscape (if it is opencast method) or huge holes underground (if it is underground mining).
- Great heaps of earth material: unwanted rock material, after the metal has been extracted from the ore, gets heaped up in tips. These are unsightly. They can be unstable and therefore dangerous. During heavy rains, a landslide is likely to occur, a catastrophe that often results to loss of life and destruction of property.
- Soil erosion: Before mining operation is carried out, the natural vegetation on or around the mining site is usually cleared up in order to give enough room to mining activities. The consequent removal of vegetation cover leaves the soil bare and, therefore, susceptible to erosion.
- Air pollution: Large-scale mineral extraction results to production of gases such as sulphur dioxide, carbon dioxide and other bad gases which are emitted to the atmosphere. These gases may bring about a green house effects and even cause acid rains.
Intervention Measures to Rectify Environmental Destruction
Suggest intervention measures to rectify environmental destruction
Different
measures are taken to check the environmental degradation (problems)
caused by mining activities. The following are some remedy measures
taken to prevent such environmental destructions:
- Governments are getting ever tougher with mining companies about damage to the environment. Sadly, in developing countries like Tanzania where much mining takes place, laws may be less strict.
- Stern controls apply to the production of wastes that may be toxic or may cause environmental damage. Safety regulations and practices must be maintained to avoid the risk of accidental release of harmful materials.
- Mine reclamation activities are undertaken gradually with the levelling of the heaps of earth material, replacement of the top soil with a fertile one and planting of trees in the mined out areas. Care must be taken to relocate streams, wildlife and other valuable resources. Quarries and opencast workings can be reclaimed by the process of filling the holes with solid wastes. The eroded bare soil can be conserved by planting trees and grasses to serve as a soil cover, which would counteract the impacts of wind, running water, rain and animals to the soil.Reclaimed land can have many uses such as agriculture, forestry, wildlife, habitation and recreation.
- Dust levels can be controlled by spraying water on roads, stockpiles and conveyors. Other steps can also be taken including filling of drills with dust collection systems, and purchasing additional land surrounding the mine to act as a buffer zone. Trees planted in these buffer zones can also minimize the visual impact of dust, from the mining operations, to local communities.
- Noise can be controlled though careful selection of equipment and insulation, and enclosures around machinery.
- The poisonous and toxic substances used in metal extraction must be treated properly before being directed into rivers and streams. Alternatively, these materials may be drained into reservoirs where they can gradually percolate deep into the soil and evaporate into the air without causing much harm to the surrounding ecosystems. In some mines, absorbent carpets are spread on the surface of the ground to trap the toxic substances contained in liquid chemicals used for mining, hence preventing these chemicals from finding their way to water bodies.
CHEMISTRY FORM THREE TOPIC 1 & 2.
CHEMISTRY FORM THREE TOPIC 3 & 4.
CHEMISTRY FORM THREE TOPIC 5 & 6.
CHEMISTRY FORM THREE TOPIC 7.
CHEMISTRY FORM THREE TOPIC 8.
O'LEVEL CHEMISTRY
CHEMISTRY STUDY NOTES, FORM FOUR.
CHEMISTRY STUDY NOTES, FORM THREE.
CHEMISTRY STUDY NOTES, FORM TWO.
CHEMISTRY STUDY NOTES FORM ONE.









No comments