Home
/
CHEMISTRY
/
CHEMISTRY FORM TWO STUDY NOTES TOPIC 5: ATOMIC STRUCTURE & TOPIC 6: PERIODIC CLASSIFICATION
CHEMISTRY FORM TWO STUDY NOTES TOPIC 5: ATOMIC STRUCTURE & TOPIC 6: PERIODIC CLASSIFICATION
TOPIC 5: ATOMIC STRUCTURE
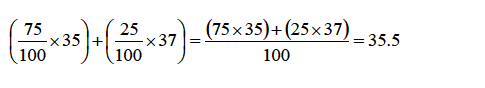
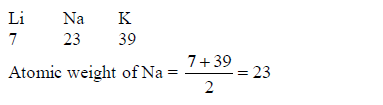

CHEMISTRY FORM TWO ALL TOPICS.
CHEMISTRY FORM TWO TOPIC 1 & 2.
CHEMISTRY FORM TWO TOPIC 3 & 4.
CHEMISTRY FORM TWO TOPIC 5 & 6.
CHEMISTRY FORM TWO TOPIC 7.
O'LEVEL CHEMISTRY
CHEMISTRY STUDY NOTES, FORM FOUR.
CHEMISTRY STUDY NOTES, FORM THREE.
CHEMISTRY STUDY NOTES, FORM TWO.
CHEMISTRY STUDY NOTES FORM ONE.
The Atom
We
learned early that matter is made up of small, indivisible particles.
Everything around us is made of extremely small particles. These
particles are either molecules or atoms. An atom is the smallest
indivisible particle of an element that takes part in a chemical change.
Atoms are the building blocks of matter. All solids, liquids and gases
are made of atoms fitted in different ways.
The
present day chemistry is built on the foundations of the Atomic Theory.
The idea that elements are made up of atoms is called the Atomic
Theory. An English chemist, John Dalton was the first to put forward the
Atomic Theory, which for most of the 19th century stated that atoms were hard, extremely small, indivisible and spherical particles like minute lead shots
Dalton Contribution to Atomic Structure
Explain Dalton contribution to atomic structure
The
Greek philosopher Democritus (460-370 BC) believed that matter was
indestructible and that it is made up of tiny particles called atoms.
Our modern understanding is based on the Atomic Theory which was put
forward by John Dalton in 1808. His theory re-introduced the ideas of
Democritus and other Greek philosophers who suggested that all matter
was infinitely divided into very small particles called atoms. These
ideas were not widely accepted at that time. They were only revived when
Dalton developed them further and experimental science was able to back
them up with practical observations.
The
Atomic Theory goes back to ancient Greeks, yet we always talk today
about Dalton's Atomic Theory. There is a good reason for this. The
reason is that, while Greeks put forward the idea that atoms exist they
did nothing more. They left the idea vague and untested. Dalton changed
this vague imaging into a set of concrete suggestions about atoms which
could be tested by experiment. This change from vagueness to precision
and experimental test justifies his claim to the theory.
Dalton’s Atomic Theory contains the following main ideas:
- Matter is made up of small, indivisible particles called atoms.
- Atoms of the same element are all exactly alike in every way and have definite weights.
- Atoms are indestructible and they cannot be created.
- Atoms of different elements have different weights and posses different properties.
- Atoms of different elements combine in small whole numbers to form ‘compound atoms’.
The Modern Concept of Dalton’s Atomic Structure
Explain the modern concept of Dalton’s atomic structure
From
the theory, it is observed that each atom has its own mass and that
chemical combination takes place between atoms and not fractions of
atoms.
Discoveries
made in the 20th century, however, showed that certain parts of the
theory must be modified. However, Dalton‟s Theory was one of the great
leaps of understanding of chemistry. It meant that we could explain many
natural processes.
Dalton's
Atomic Theory was the first step towards the formation of Modern Atomic
Theory. The Dalton's Theory has been subjected to numerous
experimentations that have led to some modifications to the theory.
However, some ideas in his theory still hold strongly to date. Some
modifications to the theory include the following:
- The atom is no longer regarded as indivisible, or the smallest particle. Particles smaller than the atom; electrons, protons and neutrons are now known. However, the atom is still the smallest particle which can take part in a chemical reaction.
- Atoms of the same element may not be all alike. Some elements have atoms with different atomic masses e.g. carbon 12 and carbon 14. These different atoms of the same element are called isotopes.
- In some few cases, atoms of different elements may have the same atomic mass. Both argon and calcium have atomic mass 40. Such atoms are called isobars.
- "The compound atoms" of Dalton are known as molecules. A molecule is the simplest particle of matter which is capable of independent existence. Evidence is available where atoms of different elements combine in large integers. An example is in organic and silicon compounds.
- Atoms are no longer regarded as indestructible. Radioactive atoms may get destroyed by spontaneous decay or by atomic fission.The atom is therefore the smallest particle of an element which is responsible for the chemical properties of that element, and which takes part in a chemical reaction.
Sub-atomic Particles
Sub-atomic Particles in an Atom
Identify sub-atomic particles in an atom
Dalton
thought that atoms were solid, indivisible particles. But, as a result
of work done mainly by Lord Rutherford, the idea has been greatly
changed in recent years. According to Rutherford, the atom consists of 3
kinds of particles - protons, neutrons and electrons. These are called
sub-atomic particles.
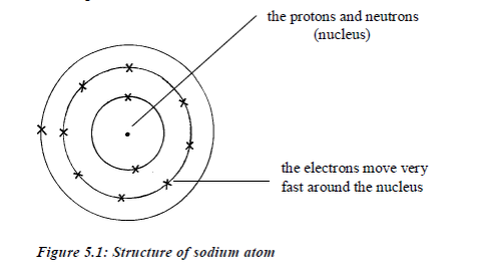
The
centre of the atom is called nucleus. The nucleus contains a cluster of
two sorts of particles, protons and neutrons. Thenucleus is very small,
occupying only about 1% of the volume of an atom. The rest of the atom
is mostly empty space, withelectrons spread out in it.

Electrons
move around the nucleus in special paths calledelectron shells
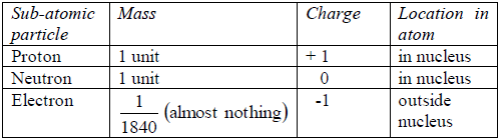
(orbits/or orbitals or energy levels). Protons andelectrons have
electric charges. Neutrons have no charges.All the particles in an atom
are very light. Their masses aremeasured in atomic mass units rather
than grams. The proton isa positively charged particle. Its mass is
about equal to that ofhydrogen atom. The neutron is has no charge, it is
neutral. Itsmass is about equal to that of hydrogen atom. The electron
isnegatively charged. Its charge is equal but opposite to the chargeon
the proton. It has a very small mass, about 1⁄1840 of the massof the
proton.
The Properties of each Particle in an Atom
Explain the properties of each particle in an atom
The properties of these particles are summarizedin the table below:
Table 5.1: Properties of sub-atomic particles

A
single atom is electrically neutral (it has no electrical charge).This
means that in any atom there must be equal numbers ofprotons and
electrons. In this way, the total positive charge onthe protons is
balanced by the total negative charge on theelectrons orbiting the
nucleus. So, the charges must cancel.
Electronic Arrangements
Electronic
arrangement refers to the manner in which electronsare arranged in an
atom. An atom contains a central nucleuscontaining protons and neutrons,
and a cluster of electronsrevolving in orbits around the nucleus. These
electrons aregrouped in shells.
A Maximum Number of Electrons in the Shells
Determine a maximum number of electrons in the shells
Bohr
(1913) put forward a theory of electron positioning whichis still
generally accepted and used until now for chemicalpurposes. Bohr's
Theory on the arrangement of electrons in anatom can be summarized as
follows:
- Electrons are in orbit around the nucleus of the atom.
- The electron orbits are grouped together in shells; a shellis a group of orbits occupied by electrons withapproximately equal energy.
- The electrons in shells distant from the nucleus havehigher energy than those in shells close to the nucleus.
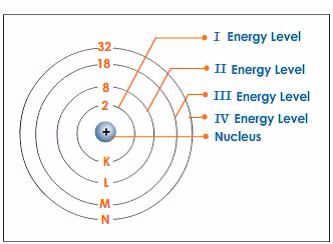
- Electrons fill the shells starting with the first shell, whichis closest to the nucleus. Shells are numbered 1, 2, 3, 4,98etc. outwards from the nucleus. The shells may berepresented by the letters K, L, M and N respectivelystarting from the nucleus.
- The maximum possible number of electrons in a shellnumbered n is 2 2n .
- The first shell can only contain up to 2 electrons. Thesecond shell can contain a maximum of 8 electrons. Thethird shell can contain up to 18 electrons.
- In the outermost shell of any atom, the maximumnumber of electrons possible is 8.
- The outer electrons of some atoms can be removed fairlyeasily to form ions.
- Chemical bonding between atoms to form moleculesinvolves the electrons in the outer shell only.
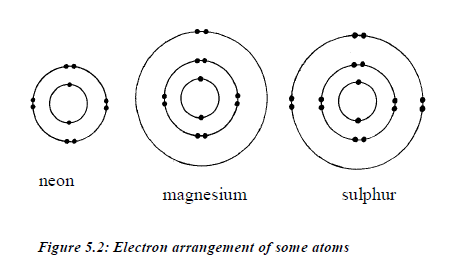
Electronic arrangement of some typical atoms is shown in figure5.2.

The
arrangement of electrons around the nucleus is also knownas electronic
configuration. This arrangement depends on themaximum number of
electrons that can occupy a shell. An atomwith 13 electrons will have
the following electronicconfiguration: 2:8:3. This means that there are 2
electrons in thefirst shell, 8 electrons in the second shell and 3
electrons in the third shell.
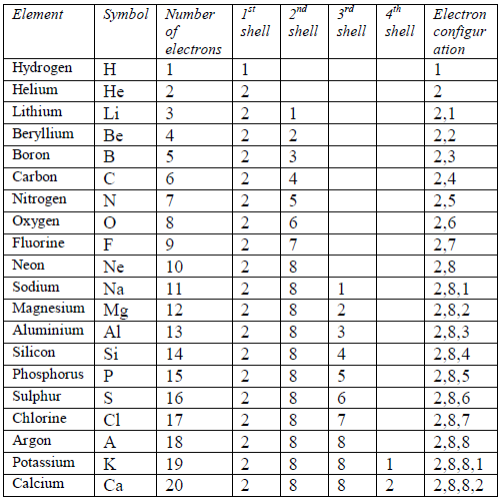
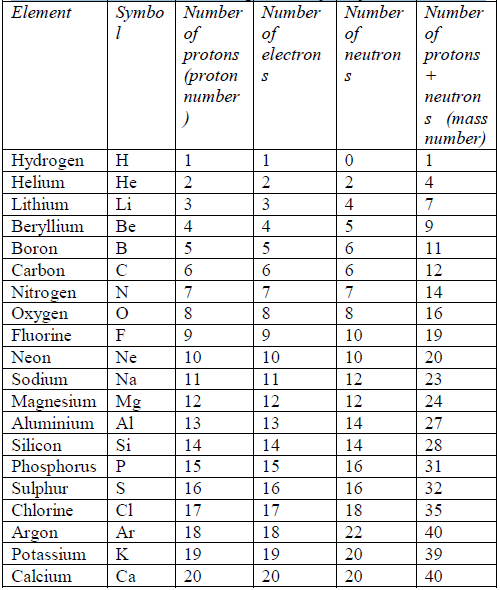
The number and arrangement of electrons in the atoms of the first 20 elements are shown in table 5.2.
Table 5.2: The electron arrangements of the first 20 elements

After
the first 20 elements, the organization of the electrons becomes
increasingly complicated. The third shell (n = 3) can be occupied by a
maximum number of 18 electrons
At
this stage, you will not be asked to work out electronarrangements
beyond element 20 (calcium), but you should beable to understand the
electronic structures involving moreelectrons (for example bromine with
the arrangement 2:8:18:7).
Energy Shell Diagrams
Draw energy shell diagrams
Energy Shell Diagram

Atomic number, Mass number and Isotope
Relationship between Atomic Number and Number of Protons
Relate atomic number with number of protons
All
atoms of one element have the same number of protons. Thisis called the
atomic number (or proton number) of that element.It is given by the
symbol Z.
No
two elements can have the same atomic number. Sodiumatoms have 11
protons. This is what makes them different fromall other atoms. Only
sodium atoms have 11 protons, and anyatom with 11 protons must be sodium
atom.
In
the same way, an atom with 6 protons must be carbon atom.Also any atom
with 7 protons must be nitrogen atom. So, youidentify an atom by the
number of protons in it. There are 109elements altogether. Of these,
hydrogen has the smallest atoms,with only 1 proton each. Helium atoms
have 2 protons each.Lithium atoms have 3 protons each, and so on up to
meitneriumatoms, which have 109 protons each. Table 5.3 shows the
first20 elements arranged according to the number of protons theyhave.
Every
atom has an equal number of protons and electrons, so theatomic number
also tells us the number of electrons in that atom.In any given atom of
an element, the number of neutrons has no effect on the identity and
properties of that particular element. It is the number of protons and
electrons that determine the identity and properties of any given
element. The number of neutrons only affects the mass, since each one of
them has the same mass as that of a proton.
Mass Number of an Atom from Numbers of Protons and Neutrons
Calculate mass number of an atom from numbers of protons and neutrons
Protons
alone do not make up all the mass of an atom. The neutrons in the
nucleus also contribute to the total mass. The mass of the electrons can
be regarded as so small that it can be ignored. As a proton and a
neutron have the same mass, the mass of a particular atom depends on the
total number of protons and neutrons present. This is called mass
number (or nucleon number). The mass number of an atom is found by
adding together the number of protons and neutrons. It is given by the
symbol A. Table 5.3 shows the mass number of the first 20 elements,
arranged in order of increasing atomic mass (mass number).
If the mass number and atomic number for any given atom are known, then its sub-atomic composition can be worked out.
The
mass number = number of protons + neutrons in an atom. Sodium atom has
11 protons and 12 neutrons, so the mass number of sodium is 23. Since
the atomic number is the number of protons only, then:
Mass
number – atomic number = number of neutrons. So, for sodium atom, the
number of neutrons = (23-11) =12. You can also take into account the
fact that, because the number of protons is always equal to the number
of electrons, then the number of electrons in sodium atom is simply 11.
The same rule can be applied to work out the sub-atomic composition of
any element.
These two relationships are useful:
- Number of electrons = number of protons = atomic number
- Number of neutrons = mass number (A) – atomic number (Z).

The Concept of Isotope
Explain the concept of isotope
Atoms
of the same element may have different numbers of neutrons. In a normal
situation, atoms of the same element will have the same number of
neutrons. However, many cases occur in which two atoms of the same
element contain the same number of protons but different numbers of
neutrons. Having equal number of protons, these atoms must also have
equal numbers of electrons. However, the differing numbers of neutrons
cause the atoms to have different mass numbers. An element showing such
properties is said to show isotopy and the varieties of the atom are
called isotopes of the element.
Therefore,
isotopy can be defined as the tendency of atoms of one element to
posses the same atomic number but different mass numbers (atomic
masses). Isotopes can be defined as atoms of the same element with the
same number of protons but different numbers of neutrons, or as „atoms
of the same element with the same atomic number but different atomic
masses‟.
The
isotopes of an element have the same chemical properties because they
contain the same number of electrons. It is the number of electrons in
an atom that decides the way in which it forms bonds and reacts with
other atoms. However, some physical properties of the isotopes are
different. The masses of the atoms differ, and therefore other
properties, such as density and rate of diffusion, also vary.
Many
isotopes (like tritium) are unstable. The extra neutrons in their
nuclei cause them to be unstable so that nuclei break spontaneously
(that is, without any extra energy being supplied), emitting certain
types of radiation. They are known as radioisotopes.
Notation for isotopes
In
order to distinguish between different isotopes of the same element in
writing symbols and formulae, a simple system is adopted. The isotope of
an element, say X will have the symbol X,AZ , where A is the mass
number of the isotope and Z is the atomic number of any atom of X. Thus,
for all isotopes of one element, Z is constant, and A varies because
there are different numbers of neutrons in the different isotopes of the
element. For example, the three isotopes of carbon are expressed as
12C6, 13C6,and 14C6. Chlorine has two isotopes: 35Cl17 and 37Cl17 .
Since A represents the total number of neutrons and protons in the
nucleus of an atom (mass number/atomic mass), and because Z is the
number of protons (atomic number), then the number ofneutrons in the
nucleus of a given isotope is given by:Number of neutrons in the nucleus
= A – Z
Relative atomic masses
As
we have seen, most elements exist naturally as isotopes.Therefore, the
value we use for the atomic mass of an element isan average mass. This
takes into account the proportions(abundance) of all the naturally
occurring isotopes. If aparticular isotope is present in high
proportion, it will make alarge contribution to the average.
Example
A
sample of chlorine gas contains 75% of the isotope 35Cl17 and 25% of
the other isotope 37Cl17 . What is the relative atomic mass of chlorine?
Solution
To work out this problem, simply multiply the mass number ofeach isotope with the abundance and sum up the products thus:
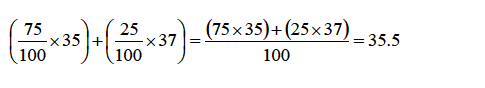
This
average value for the masses of atoms of an element isknown as the
relative atomic mass (Ar).Therefore, the relative atomic mass of
chlorine is 35.5 (i.e., Ar =35.5).
TOPIC 6: PERIODIC CLASSIFICATION
Constructing
the modern periodic table has been a major scientific achievement. The
first steps towards working out this table were taken long before anyone
had any idea about the structure of atoms. The number of elements
discovered increased steadily during the 19th century. Chemists began to
find out patterns in their properties.
The Law of Triads
In
1817, the German scientist Johann Dobereiner noticed that calcium,
strontium and barium had similar properties, and that the atomic weight
of strontium was halfway between the other two. He found the same
pattern with chlorine, bromine and iodine and also with lithium, sodium
and potassium. So, he put forward the law of Triads: “If elements are
arranged in groups of three in order of increasing atomic weights,
having similar properties, then the atomic weight of the middle element
is the arithmetic mean of the atomic weights of the other two elements”,
E.g.
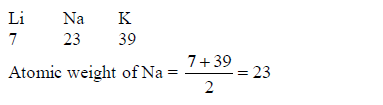
The
following are examples of Dobereiner's triads:(Lithium, Sodium and
Potassium)(Calcium, Strontium and Barium)(Chlorine, Bromine and Iodine)
and(Iron, Cobalt and Nickel)
The Law of Octaves
In
1863 John Newlands, an English chemist noted that there were many pairs
of similar elements. In each pair, the atomic weights differed by a
multiple of 8. So, he produced a table with the elements in order of
increasing atomic weights, and put forward the Law of Octaves: “If
elements are arranged in order of their increasing atomic weights, the
properties of the 8th element, starting from a given one, are a kind of
repetition of the first element”.
This finding was comparable to the 8th note of music, hence the use of the word "octave".
This
was the first table to show a periodic or repeating pattern of
properties. But it was not widely accepted because there were too many
inconsistencies. For example, he put copper and sodium in the same
group, even though have very different properties. Also iron was placed
in the same group as oxygen and sulphur.
The Periodic Law
Dmitri
Mendeleev was born in Siberia, Russia, in 1834. By the time he was 32,
he was a professor of Chemistry. In 1869 Mendeleev advanced the work
done by Newlands and contributed very useful new ideas. He began by
listing all the known elements in order of increasing atomic mass. He
spotted that elements with similar properties appear at regular
intervals or periods down the list. His findings were the basis for the
Periodic Law: “The properties of elements are a periodic function of
their atomic masses”.
Mendeleev
placed similar elements into groups. He realized that not all elements
had been discovered. So he left gaps for new ones in the correct places
in his table. He also swapped the order of some elements to make them
fit better. He predicted the properties of the missing elements from the
properties of the elements above and below them in the table. He also
listed separately some elements which did not appear to fit into any
group i.e. iron, cobalt, nickel, etc.
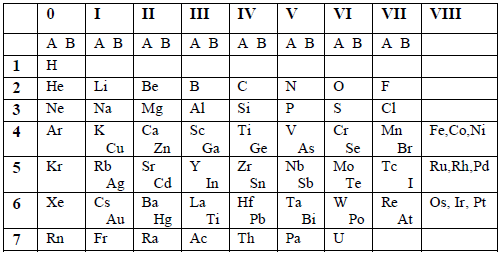
Table 6.1: Mendeleev’s short form of the Periodic Table

The
table had 9 vertical columns which he called Groups. The groups were
numbered from 0 to 8. The elements in group 0 were not known by then,
but were discovered later on. Groups 1 to 7 were subdivided into A and B
subgroups. Group 0 included the transition elements. Noble gases were
later placed in group 0.
There
were 7 horizontal rows which he called periods. All vacant positions in
the table stood for new elements yet to be discovered.
Usefulness of Mendeleev's classification
- The table summarized a large amount of information about the elements based on their chemical properties.
- The table was very useful in predicting the existence and properties of undiscovered elements, for which gaps had been left in the table.
- The table was also used in checking relative atomic masses of elements.
Limitations of Mendeleev's classification
- In three cases, pairs of elements had to be included in one group based on inverse order of their atomic weights so as to fit into groups of elements having similar properties. These pairs were argon (39.9) and potassium (39.1), cobalt (58.9) and nickel (58.9); plus tellurium (127.5) and iodine (126.9). This difficulty was resolved when the basis of classification was based on the atomic number instead of the atomic mass.
- The elements that were placed in group VIII formed an incompatible mixture.
- The placing of two different families in one group e.g. K and Cu; Ca and Zn, etc.
The
periodic table is the chemists map. It helps you understand the
patterns in chemistry. Today we take it for granted. But it took
hundreds of years, and work of hundreds of chemists, to develop.
The
Modern Periodic Table is similar to that of Mendeleev, but contains
several improvements. Elements are arranged in order of atomic number
instead of atomic mass. This means that elements no longer have to swap
places to fit correctly. Many new elements have been discovered and
slotted into the spaces left by Mendeleev. Also metals and non-metals
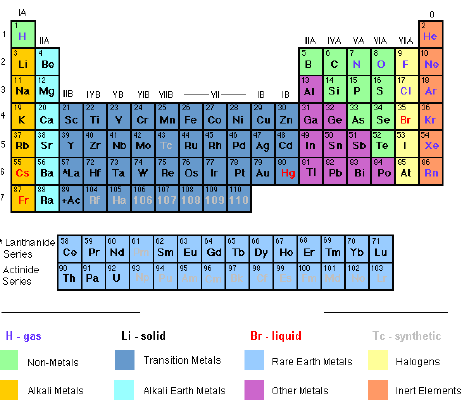
are clearly separated. The Modern Periodic Table is shown in Figure 6.1.
Figure 6.1: The Modern Periodic Table

The
long form of the periodic table is the commonly used form of the
periodic table. The elements in the table are arranged based on their
atomic weights, starting from hydrogen (1), helium (2), lithium (3),
beryllium (4) and so on. The elements appear in vertical columns and
horizontal rows.
The
vertical columns in the table are called Groups, numbered I, II, III,
IV, V, VI, VII and 0, which is also known as group VIII. Group I
contains the elements lithium (L), sodium (Na), rubidium (Rb), caesium
(Cs) and francium (Fr). Group II consists of elements starting from
sodium (Na) down to radium (Ra). Some of the groups have special names.
- Group I is often called the alkali metals.
- Group II the alkaline earth metals.
- Group VII the halogens.
- Group 0 the noble gases.
The
transition metals (or elements) form a separate block in the middle of
the periodic table between group II and III. The atoms of these elements
have more complicated electron arrangements. Note that the group
contains many common metals such as iron (Fe), Nickel (Ni), copper (Cu),
and Zinc (Zn). One of the interesting properties of these elements is
that they form coloured compounds.
Main features of the Modern Periodic Table
- The elements in the table are placed in order of their atomic numbers instead of their atomic masses.
- There are a total of 18 groups and 7 periods.
- There are 5 blocks of similar elements in the periodic table as shown in figure 6.2.
- The normal (non-transition) elements (groups 1-7) have their outermost shells incomplete, meaning that they can allow additional electrons to enter into their outermost orbital (valency shell). But each of their inner shells is complete.
- The transition metals have their outermost as well as their penultimate (second last) shells incomplete.
- Elements of group 0 (noble gases) have their shells complete. These elements show little reactivity. That is why they wereonce called „inert‟ gases because they are very unreactive; or „rare gases‟ because they were rarely found.
- Gaps left by Mendeleev for undiscovered elements (now occupied by the transition elements and the noble gases) have been filled by the respective elements following their discovery. Man-made elements have also found a place in the periodic table.
- Metals have been clearly separated from non-metals. Metalloids or semi metals (poor metals) have also been included. Metalloids are elements whose properties are intermediate between metals and non-metals. They include boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb) and tellurium (Te). In some publications, germanium and antimony are usually classed as poor metals and the rest as non-metals.

Periodicity
The Concept of Periodicity
Explain the concept of periodicity
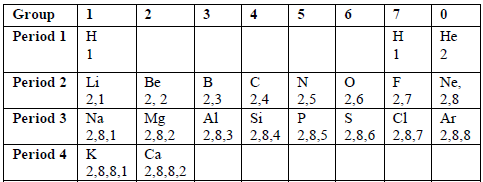
Consider the electronic configuration of the first twenty elements of the periodic table shown in the table below.
Table 6.3: Electronic configurations of the first 20 elements

You
will notice that elements in the same vertical columns (groups) have
the same number of electrons in the outermost shells of their atoms.
Because the outer electrons determine the chemical properties of an
element, then the elements in each period tend to resemble each other
closely in chemical behaviour. For instance, the noble gases, He, Ne and
Ar show a chemical inertness which is characterised by the stable outer
electron octet or duplet. Due to this reason, the compounds of the
noble gases with other elements have not been found.
Attempts
to classify elements by arranging them in order of increasing atomic
weights shows that the properties of elements were periodic. This means
elements with similar or comparable properties appear after a certain
specific interval in a given arrangement. The occurrence of successive
groups of elements showing strong chemical similarity in this way is
called periodicity.
Therefore,
periodicity is the repetition of similar chemical properties of
elements after a certain specific interval in a given arrangement. The
repetition in properties is due to repetition of similar electronic
configuration of outermost shells of elements after certain intervals.
General Trends
This
refers to change in some properties of elements across the periods and
down the groups in the periodic table. These trends become more obvious
if we leave aside the noble gases in Group 0. In this case, we shall
concentrate our efforts on variations in the most important properties
of the elements only. The following is a summary of the change in some
properties of elements down the groups and across the periods.
The Change in Properties of Elements Across the Periods
Explain the change in properties of elements across the periods
Atomic and ionic size
The
sizes of atoms and ions may be given in terms of atomic radius and
ionic radius units respectively. The number of shells an atom or ion
posses and the nuclear charge determines the size of an atom or ion.
This is how the two properties vary along the period and down the group:
Atomic size
Along the period:
Considering the normal elements only, the size of the atoms decrease
from left to right across the period. This is because as atomic number
increases across the period, the nuclear charge (due to increasing
protons) increases and electrons in shells are pulled closer to the
nucleus.
Ionic size
- Positive ions (cations):Across the period; The ionic size does not change, i.e. remains the same, as you move across the period from either direction.
- Negative ions (anions):A negative ion is larger compared to the corresponding neutral atom because on forming an ion, one or more electrons are added to the atom. The added electron(s) is/are repelled by the electron(s) already present in the outermost shell, hence leading to an increase in the size of an atom, even though no new shell is formed.Down the group and along the period: Ionic size increases down the group, and along the period, i.e. from left to right.
Atomic radii (singular: radius)
Along the period: In the period, atomic radii decrease from left to right with increase in the atomic number.
Electronegativity
Electronegativity
is the tendency of an atom to attract the shared pair of electrons
towards itself in a molecule. The electronegativity values of elements
in group 0 (inert gases) is zero.
Along the period: Electronegativity increases while moving across the period from left to right in the periodic table.
Metallic character (or electropositivity)
Electropositivity
is the tendency of an element to lose the valency electron(s) and
donate the same to other elements (usually non-metallic elements). This
process occurs during the formation of new substances e.g. molecules and
compounds. Literally, such reactions occur between metals and
non-metals whereby metals donate electrons and non-metals receive these
electrons. So, metals are electropositive elements while non- metals are
electronegative elements.
Along the period:
Generally, metallic character decreases along the period from left to
right.The gradation in metallic properties across the period is as
follows: Metals → poor metals → metalloids → non-metals → noble gases
Chemical reactivity
Reactivity is the tendency of an element to lose or gain electrons in a chemical reaction.
Along the period: For metals, the reactivity decreases from left to right in a period while it increases for non-metals.
Ionization Energy or Ionization Potential (I.E or I.P)
This
refers to the minimum amount of energy required to remove the most
loosely bound electron from an isolated atom or ion in its gaseous
state. The smaller the value of ionization energy, the easier it is to
remove the electron from the atom.M(g) →M+(g) + e-
Along the period: It increases along the period from left to right with the increase in atomic number.
Electron affinity (Ea):
This
is just opposite to I.E. It is defined as the amount of energy released
when an extra electron is added to an isolated neutral atom in its
gaseous state.
Along the period: The value increases along the period from left to right.
Density and melting point
The
density of a substance is the ratio of its mass to its volume, while
the melting point is the temperature at which a solid substance turns
into liquid at standard atmospheric pressure.
- Density-Across the period: Densities decrease across the period from left to right.
- Meting point-Across the period: Melting points of elements decrease across the period from left to right.
The Change in Properties of Elements Down the Groups
Explain the change in properties of elements down the group
Atomic and ionic size
- Atomic size-Down the group: Atomic size increases as you move down the group.
- Ionic size- Positive ions (cations)-Down the group: On descending the group, the nuclear charge increases and the number of shells increase by one at each step so, the ionic size also increases. A positive ion is smaller than the corresponding neutral atom because on forming the ion, the metal atom loses both the valency electron(s) and the outermost shell. Valency electron(s) refer(s) to the electron(s) in the outer-most shell of an atom. Any further removal of electron(s) from the ion will decrease the ionic size further.Negative ions (anions)-Down the group and along the period: Ionic size increases down the group, and along the period, i.e. from left to right.
Atomic radii (singular: radius)
Atomic radius is the distance from the centre of the nucleus to the outermost shell (valency shell). Down the group: Atomic radii of elements increase down the group with increase in atomic size.
Electronegativity
Down the group: Electronegativity decreases while moving downwards in a group.
Metallic character (or electropositivity)
Down the group: Metallic character (electropositivity) increases down the group
Ionization Energy or Ionization Potential (I.E or I.P)
Down the group: It decreases gradually down the group.
Why
is there a decrease in I.E as you go down the group? This is because
electrons are held in their shells by their attraction to the positive
nucleus, and as you go down the group, the size of the atom increases
(increasing atomic radius). So, the outermost electron(s) of an atom
gets further and further away from the attraction or pull of the
positive nucleus, hence requiring little energy to remove from the atom.
Electron affinity (Ea)
Down the group: The value of electron affinity decreases down the group.
Density and melting point
- Density-Down the group: Densities of elements increase down the group.
- Meting point-Down the group: Melting points of elements decrease down the group as the elements become less metallic in nature.
Electronic Configuration to Locate the Positions of Elements in Periodic Table
Use electronic configuration to locate the positions of elements in periodic table
The
modern periodic table is based on electronic configurations of the
elements. Look at table 6.3 and study the electronic configurations of
the first twenty elements and where they are placed in the periodic
table.
Beryllium, magnesium and calcium have two electrons in the outer shell. These elements are in Group 2.
This
pattern continues to Group 3, Group 4 and so on. The group number in
the periodic table is the same as the number of electrons in the
outermost shell. The halogens are the elements in Group 7. Bromine is
one of the halogens. How many electrons does each bromine atom have in
its outer shell?
As
we move down each group, the number of shells increases by one at each
step. Each atom of an element has one complete shell than the one above
it.
As
we move across each period, the outer shell is being filled by one
electron at each step. Certain electronic configurations are found to be
more stable than others are. The noble gases at the end of each period
have full outer shells. They have stable duplet (2 electrons) or octet
(8 electrons) in their outermost shells. This makes them more difficult
to break up, and this fits well with the fact that they are so
unreactive.
The
outer electrons of an atom are mainly responsible for the chemical
properties of an element. Therefore, elements in the same group will
have similar chemical properties.
CHEMISTRY FORM TWO ALL TOPICS.
CHEMISTRY FORM TWO TOPIC 1 & 2.
CHEMISTRY FORM TWO TOPIC 3 & 4.
CHEMISTRY FORM TWO TOPIC 5 & 6.
CHEMISTRY FORM TWO TOPIC 7.
O'LEVEL CHEMISTRY
CHEMISTRY STUDY NOTES, FORM FOUR.
CHEMISTRY STUDY NOTES, FORM THREE.
CHEMISTRY STUDY NOTES, FORM TWO.
CHEMISTRY STUDY NOTES FORM ONE.









Great job for publishing such a nice article. Your article isn’t only useful but it is additionally really informative. Thank you because you have been willing to share information with us. If anyone looking for the a level chemistry tuition services, nanyangacademics is the best choice.
ReplyDeleteThis particular is usually apparently essential and moreover outstanding truth along with for sure fair-minded and moreover admittedly useful igcse tuition chemistry kuala lumpur
ReplyDelete